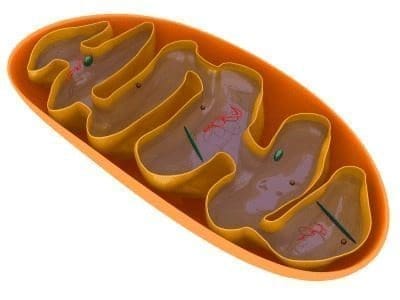
Antioxidantes Mitocondriales Por cuanto el daño oxidativo es parte de la patogenia de la diabetes, es lógico que los antioxidantes tengan potencial terapéutico.
Los compuestos naturales tales como el atocoferol, el ascorbato, la coenzima Q y el ácido lipoico, no tienen una definida acción, aunque administrados en dosis altas hayan demostrado efectividad en otras entidades.
Experimentalmente demuestran que tanto la vitamina E como la coenzima Q protegen a la mitocondria contra la toxicidad del O2 y que la acumulación de los mismos por el organelo aislado aumentan la eficacia de su acción44.
Se ha comprobado que el compuesto alquiltrifenilfosfonio atraviesa las dos capas de la membrana mitocondrial, sin requerir transportadores o moléculas de acoplamiento45.
Así mismo el metil-trifenil-fosfonio no se acumula y es depurado. Ha sido cuantificado en otros tejidos: cerebro, miocardio e hígado en el ratón y desaparece de la circulación una vez suspendida su administración.
Es importante decir que las concentraciones de estos componentes en la mitocondria, es centenares de veces superior, de las cantidades presentes en el citosol.
Hasta el presente diversos protocolos estudian in vivo los compuesto antioxidantes en modelos experimentales con daño mitocondrial.
Conclusiones
En conclusión, el propósito de esta revisión ha sido presentar un aspecto optimista para quienes ven a la diabetes como una enfermedad con un pronóstico cada vez menos próximo a la curación y a la prevención.
Pero el progreso no puede detenerse.
Gracias a que el mundo científico está cada vez más concentrado en la investigación básica y a la aplicación de sus resultados, diversas oportunidades se pueden ofrecer al enfermo.
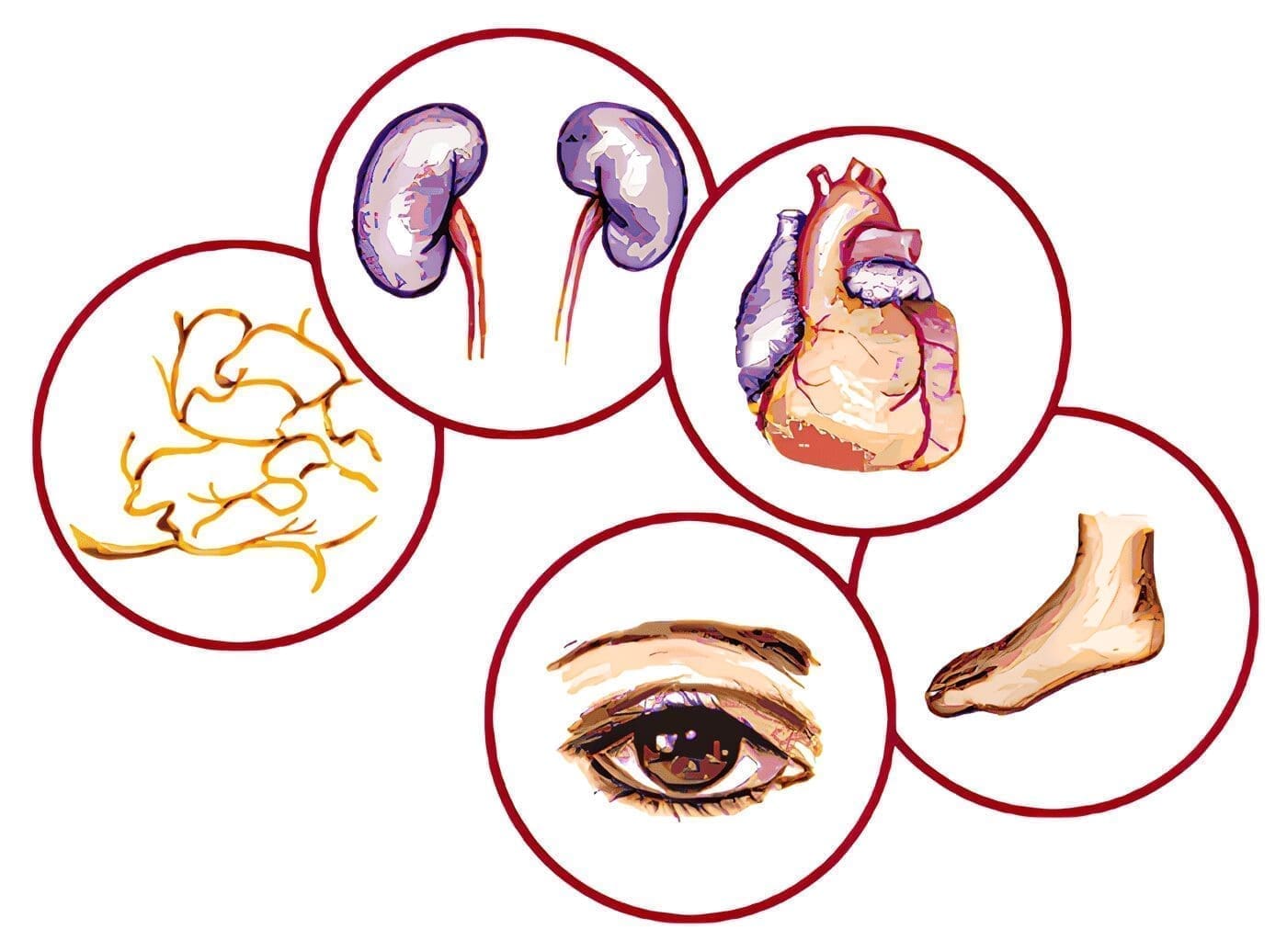
Como resumen se analizaron los estudios epidemiológicos, la patogénesis, las bases químicas y los mecanismos biomoleculares. Llevan al daño vascular prematuro en la intolerancia a la glucosa. Cuando aún no se ha detectado en el individuo una glicemia que lo catalogue como diabético tipo 2, a saber:
- Las alteraciones micro vasculares en la intolerancia a la glucosa en dos estudios controlados.
- El estrés oxidativo por la hiperglicemia y sus consecuencias sobre el daño celular.
- Los mecanismos en las vías del poliol y la hexosamina en la generación de radicales de oxígeno tóxicos.
- La participación y modo de acción de los productos de glicosilación avanzada (AGEs) en el daño micro vascular.
- La activación de la proteína-cinasa C por el diacil-glicerol en la alteración de la curva de tolerancia a la glucosa y los mecanismos que conducen a una serie de eventos bio-moleculares.
- La participación de la mitocondria en la producción de O2 y la utilización de antioxidantes en este proceso.
- Las posibles intervenciones y progresos en el período anterior a la aparición de la diabetes 2.
Bibliografía
- 1. Harris MI, Klein R, Wellborn TA, Knuiman MW: Onset of NIDDM occurs at least 4-7 years before clinical diagnosis. Diabetes Care 15:815-819, 1992
- 2. Diabetes Care 20: 1183-99, 1997
- 3. Reaven G. Role of insulin resistance in human disease. Diabetes 37:1595-1607, 1988
- 4. DeFronzo, RA, Ferrannini E. Insulin Resistance. A multifaceted syndrome responsible for NIDDM, obesity, hypertension, dyslipidemia and atherosclerotic cardiovascular disease. Diabetes Care14:173-194, 1991
- 5. Alberti KGMM, Zimmet PZ for the WHO consultation. Definition, diagnosis and classification of diabetes mellitus and its complications. Diabetes Med 15:539-553, 1998
- 6. Sinha R, Fisch G, Teague B, Tamborlane WW et al. Prevalence of impaired glucose tolerance among children and adolescents with marked obesity. N Engl J Med 346:802-810, 2002
- 7. Diabetes Prevention Program Research Group. Reduction in the incidence of type 2 diabetes with lifestyle intervention or metformin. The New Engl J of Med 346:393-403, 2002
- 8. Mykkanen L, Haffner SM, Kuusisto J, et al. Microalbunminuria precedes the development of NIDDM. Diabetes 43:552 – 557; 1994
- 9. Klein R, Barrett-Connor EL, Blunt BA, Wingard DL. Visual impairment and retinopathy in people with normal glucose tolerance, impaired glucose tolerance, and newly diagnosed NIDDM. Diabetes Care 14:914-918,1991
- 10. Pan XR. Hu YH, Li GW, Liu PA, Bennett PH, Howard BV: Impaired glucose tolerance and its relationship to ECG-indicated coronary heart disease and risk factors among Chinese. Da Qing IGT and diabetes study. Diabetes Care 16:150-156, 1993
- 11. Metcalf PA, Baker JR, Scragg RK, Drson E, Scott AJ, Wild CJ: Microalbuminuria in a middle-aged workforce: effect of hyperglycemia and ethnicity. Diabetes Care 16:1485-1493, 1993
- 12. Mogensen C: Microalbuminuria predicts clinical proteinuria and early mortality in maturity-onset diabetes. N Engl J Med 310:356-360, 1984
- 13. Wei M, Gaskil SP, Haffner SM & Stern, MP. Effects of diabetes and level of glycemia on all-cause and cardiovascular mortality. The San Antonio Heart Study. Diabetes Care 7, 1167-1172, 1998
- 14. Ebara T. et al. Delayed catabolism of apob-48 lipoproteins due to decreased heparan sulfate proteoglycan production in diabetic mice. J Clin Invest 105, 1807-1818, 2000
- 15. Ginsberg HN. Insulin resistance and cardiovascular disease. J Clin Invest 106, 453-458, 2000
- 16. Wilkin TJ: The accelerator hypothesis: weight gain as the missing link between type 1 and type 2 diabetes. Diabetologia 44:914-922, 2001
- 17. Libman I, Arslanian S: Type 2 diabetes in childhood: The American perspective. Horm Res 59 (Suppl.1):69-76, 2003
- 18. Weets I, Van Autreve J, Van der Auwera BJ et al. Male-to-female excess in diabetes diagnosed in early adulthood is not specific for the immune-related form, nor is it HLA-DQ restricted: possible relation to increased body mass index. Diabetologia 44:40-47,2001
- 19. Clausen JO, Borch-Johnsen K, IbsenH et al: Insulin sensitivity index, acute insulin response and glucose effectiveness in a population-based sample of 380 young healthy Caucasians: analysis of the impact of gender, body fat, physical fitness and lifestyle factors. J Clin Inves 98:1195-1209, 1996
- 20. Kibirige M, Metcalf B., Renuka R. Et al: Testing the accelerator hypothesis. Diabetes Care 26:2865-2870,2003
- 21. Ingawa A, Hanafusa T, Miyagawa JI, Matszawa Y: A novel subtype of Type 1 diabetes mellitus characterized by a rapid onset and an absence of diabetes-related antibodies. N Engl J Med 342:301-07, 2000
- 22. Horton V, Stratton I, Bottazzo GF et al: Genetic heterogeneity of autoimmune diabetes: age of presentation in adults is influences by HLA DRB1 and DQB1 genotypes (UKPDS43). UK Prospective Diabetes Study (UKPDS) Group. Diabetologia 42, 608-16, 1999
- 23. Ross R: The pathogenesis of atherosclerosis: a perspective for the 19902. Nature 362:801-809, 1993
- 24. Hu FB, Stampfer MJ: Is type 2 diabetes mellitus a vascular condition? Arterioescler Thromb Vasc Biol 23:715-1716, 2003
- 25. Jiang ZY et al. Characterization of selective resistance to insulin signaling in the vasculature of obese Zucker (fa/fa) rats. J Clin Invest 104, 447-457, 1999
- 26. Singleton RJ, Smith GA, Russell JW, Feldman EL: Microvascular complications of impaired glucose tolerance. Diabetes 52:2867-2873, 2003
- 27. Brownlee M: Biochemistry and molecular cell biology of diabetic complications. Nature 414:813-820, 2001
- 28. Williamson JR et al: Hyperglycemic pseudohypoxia and diabetic complications. Diabetes 42,801-813, 1993
- 29. Greene DA, Arezzo JC & Brown MB: Effect of aldose reductase inhibition on nerve conduction and morphometry in diabetic neuropathy. Zenarestat study group. Neurology 53, 580-591, 1999
- 30. Thornalley PJ: The glyoxalase system: new developments towards functional characterization of a metabolic pathway fundamental to biological life. Biochem J 269, 1-11, 1990
- 31. Degenhardt TP, Thorpe SR & Baynes JW: Chemical modification of proteins by methylglyoxal. Cell Mol Biol. 44, 1139-1145, 1998
- 32. Tanaka S, Avigad G, Brodsky B & Eikenberry EF: Glycation induces expansion of the molecular packing of collagen. J Mol Biol 203, 495-505, 1988
- 33. Haitoglou CS, Tsilibary EC, Brownlee M & Charonis AS: Altered cellular interactions between endothelial cells and nonenzymatically glucosylated laminin/type IV collagen. J Biol Chem 267,12404-12407, 1992
- 34. Neeper M et al: Cloning and expression of RAFE: a cello surface receptor for advanced glycosylation end products of proteins. J Biol Chem 267, 14998-15004, 1992
- 35. Yan SD et al: Enhanced cellular oxidant stress by the interaction of advanced glycation end products with their receptors/binding proteins. J Biol Chem 269, 9889-9897, 1994
- 36. IshiiH et al: Amelioration of vascular dysfunctions in diabetic rats by an oral PCK beta inhibitor. Science 272, 728-731, 1996
- 37. Kuboki K et al: Regulation of endothelial constitutive nitric oxide synthase gene expression in endothelial cells and in vivo a specific vascular action of insulin. Circulation 101, 676-681, 2000
- 38. Williams B, Gallacher B, Patel H & Orme C: Glucose-induced protein kinase C activation regulates vascular permeability factor mRNA expression and peptide production by human vascular smooth muscle cells in vitro. Diabetes 46, 1497-1503, 1997
- 39. Koya D et al: Characterization of protein kinase C beta isoform activation on the gene expression of transforming growth factor-beta, extracelular matrix components, and prostanoids in the glomeruli of diabetic rats. J Clin Invest 100, 115-126, 1997
- 40. Feener EP et al: Role of protein kinase C in glucose and angiotensin II-induced plasminogen activator inhibitor expression. Contrib Nephrol 118, 180-187, 1996
- 41. Klom-Litty V, Sauer U, Nerlich A, Lehmann R & Schleicher ED: High gucose-induced transforming growth factor betal production is mediated by the hexosamine pathway in porcine glomerular mesangial cells. J Clin Invest 101, 160-169, 1998
- 42. Du et al: Hyperglicemia-induced mitochondrial superoxide overproduction activates the hexosamine pathway and induces plasminogen activator inhibitor-1 expression by increasing Sp1 glycosylation. PNAS 97 No. 22,12222-12226,2000
- 43. Nikishikawa T et al: Normalizng mitochondrial superoxide production blocks three pathways of hyperglycemic damage. Nature 404, 787-790, 2000
- 44. Castano L, Eisenbarth GS: Type 1 diabetes: A chronic autoimmune disease of human, mouse, and the rat. Annu Rev Immunol 8:647-79, 1990
- 45. Atkinson MA, Maclaren NK. The pathogenesis of insulin dependent diabetes. N Engl J Med 331:1428-36, 1994
- 46. LaPorte RE, Tajima N, Kerblom HK, et al. Geographic differences in The risk of insulin-dependent diabete4smellitus: the importance of registries. Diabetes Care 8(suppl): S101-07, 1985
- 47. Anonymous. EURODIAB ACE Study Group. Variation and trends in incidence of childhood diabetes in Europe. Lancet 355:873-76, 2000
- 48. Onkamo P, Vonnen S, Karvonen M, et al: Worlwide increase in incidence to type 1 diabetes – the analysis of the data on published incidence trends. Diabetologia 42:1395-1403, 1999
- 49. Caux C, Vanbervliet B, Massacrier C, et al: CD34+ hematopoietic progenitors from human cord blood differentiate along two independent dendritic cell pathways in response to GM-CSF+TNFa. J Exp Med 184:695-706, 1996
- 50. Banchereau J, Steinman RM. Dendritic cells and the control of immunity. Nature 392:245-52, 1998
- 51. Thomas R, Lipsky PE. Presentation of self peptides by dendritic cells: possible implications for the pathogenesis of rheumatoid arthritis. Arthritis Rheum 39:183-190, 1996
- 52. Todd JA. From genome to aetiology in a multifactorial disease, type 1 diabetes. BioEsays 21164-74, 1999
- 53. Buzzetti R, Quattrocchi CC, Nistico L. Dissecting the genetics of type 1 diabetes: relevance familial clustering and differences in incidence. Diabetes Metab Rev 14:111-28, 1998
- 54. Lernmark A, Ott J. Sometimes it is hot, sometimes it is not. Nat Genet 19:213-14, 199
- 55. Ellis TM, Atkinson MA. Early infant diets and insulin-dependent diabetes. Lancet 347:1464-65, 1996
- 56. Dahlquist GG. Viruses and other perinatal exposures as initiating events for beta-cell destruction. Ann Med 29:413-17, 1997
- 57. Knip M, Akerblom HK. Environmental factors in the pathogenesis of type I diabetes mellitus. Exp Clin Endocrinol Diabetes 107(Supp):S93-100, 1999
- 58. Eisenbarth GS. Type 1 diabetes mellitus. A chronic autoimmune disease. N Engl J Med 314:1360-68, 1986
- 59. Serreze DV, Ottendorfer EW, Ellis TM et al. Acceleration of type 1 diabetes by a coxsackievirus infection requires a preexisting critical mass of autoreactive T-cells in pancreatic islets. Diabetes 49:708-11, 2000
- 60. Zimmet PZ, Tuomi T, Macaky IR et al. Latent autoimmune diabetes mellitus in adults (LADA): the role of antibodies to glutamic acid decarboxylase in diagnosis and predictin of insulin dependency. Diabet Med 11:299-303, 1994
- 61. Turner R, Stratton I, Horton V et al. UKPDS 25: autoantibodies to islet-cell cytoplasma and glutamic acid decarboxylase for prediction of insulin requirement in type 2 diabetes. UK Prospective Diabetes Study Group. Lancet 350:1288-93, 1997
- 62. Knip M, Douek IF, Moore et al. Safety on high-dose nicotinamide: a review. Endit Group. European Nicotinamide Diabetes Intervention Trial. Diabetologia 43:1337-45, 2000
- 63. DPT-1 Study Group. The Diabetes Prevention Trial-Type 1 diabetes (DPT-1): implementation of screening and staging of relatives. Transplant Proc 27:3377,1995