La inmunotolerancia inducida por el uso de la vía oral y sublingual, tiene una importante aplicación en el tratamiento de desensibilización de procesos alérgicos, (46, 51, 58, 67, 68, 70-77, 89, 102, 103, 110, 112, 138, 141, 147, 155, 173) dermatitis por contacto a níquel, cromo y estaño (4, 12, 45, 173).
Es en este campo es donde se ha acumulado más experiencia investigativa y se ha logrado pasar sutilmente de la fase puramente experimental a la aplicación en el humano (173, 174-176).
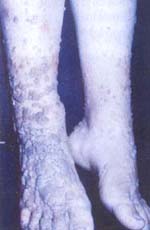
La dermatitis por contacto a níquel es las más estudiada, la utilización de pequeñas dosis de este metal, por vía oral a un animal impiden la inducción intencional de esta patología (4-12, 45,102).
Debemos recordar que el estaño y el níquel son metales muy conocidos por su gran capacidad de producir dermatitis por contacto en el humano cuando son usados en adornos de fantasía de uso femenino, como aretes, cadenas, pulsos, etc.
La experimentación en este tipo de enfermedades ha arrojado resultados muy interesantes, además de inducirse tolerancia específica, ésta es persistente, con períodos de observación superiores a los dos años (45).
En pediatría, la alergia a la leche de vaca (46, 52, 65, 74, 137, 112) y al huevo (1, 28, 51, 112, 137) son las más exploradas. En trabajos experimentales con ratones, administrándoles inicialmente caseína bobina (S1CN) como alimento y luego al inocular los animales por vía parenteral con esta proteína, no se evidenció ningún tipo de respuesta inmunitaria, por el contrario hubo tolerancia plena a la S1CN (46).
Algunos investigadores sugieren que la inmunotolerancia oral se da por una anergia inducida en LT (51).
Como lo demuestran diversas investigaciones, la administración de extracto de polen de gramíneas por vía oral produce un estado de tolerancia, muy útil en los procesos alérgicos respiratorios ocasionados por pólenes estacionales, de esta manera, se suministra el tratamiento de inmunoterapia sublingual, nasal u oral con estos pólenes antes de la estación en la cual se producen; cuando llega la estación respectiva con su polinización característica no se observa la recaída de la enfermedad alérgica, por tanto, nos indica que el paciente ha sido desensibilizado eficazmente (58, 156, 158, 159, 162-164, 166, 169, 170, 173).
Relacionado con la alergia a pólenes existe un trabajo realizado con el antígeno Parietaria judaica administrado por vía sublingual (128); se realizó en 40 pacientes alérgicos que padecían rinoconjuntivitis y asma bronquial secundaria a este alérgeno. Se procedió de la siguiente manera:
• 20 pacientes como control.
• 20 tratados con el alergeno Parietaria judaica por vía sublingual.
En los pacientes tratados con el alergeno no se presentaron efectos colaterales y durante la estación en la cual predomina este polen en el ambiente, ellos no tuvieron recaídas de su rinoconjutivitis (128).
En la desensibilización de los procesos alérgicos se deben administrar dosis bien estandarizadas, requiriendo de múltiples administraciones del alergeno, sujetas a las recomendaciones del fabricante o al estudio que haga el investigador para cada alergeno en particular (54, 67, 113, 114, 135).
Trabajos similares se han realizado en alergia alimentaria al huevo. Animales previamente sensibilizados con este producto por vía parenteral, se les indujo tolerancia al administrarles por vía oral clara de huevo obteniéndose como resultado una desensibilización efectiva contra esta proteína (1, 28, 51, 54, 65, 112, 137).
En este caso también es muy importante las dosis e intervalos de administración pues de esto depende el hecho de no pasar de un estado de tolerancia a una sensibilización o provocación de inmunidad ante un alergeno (54).
Lo que respecta a alergia alimentaria uno de los trabajos más interesantes fue el llevado a cabo con proteínas de leche de soya (68), descrito a continuación:
- Se seleccionaron cerditos de dos, ocho y trece semanas de edad ocho en cada grupo, fueron previamente inmunizados intraperitonealmente con proteínas de soya con adyuvante Quica. Luego se alimentaron durante doce semanas con leche de soya.
- Se seleccionó un grupo control al cual se inyectó por vía intraperitoneal (VIP) con adyuvantes Quica únicamente.
Todos los animales alimentados con leche de soya demostraron respuesta IgG a esta proteína, esto indicó que la alimentación con proteínas de soya no indujo tolerancia a la misma, después de haber sido previamente inmunizado el animal.
La experiencia se repitió administrando la proteína de soya antes de la sensibilización ip, se obtuvo un estado de tolerancia que se demostró cuando los animales fueron posteriormente inmunizados por vip.
El hallazgo más importante de esta investigación fue que al inmunizar por vip con la proteína de la leche de soya, sólo se obtuvo tolerancia en los animales que habían sido destetados después de la semana 12 de su nacimiento, es decir, en aquellos que tenían un sistema inmunitario de mucosas más maduro y en condiciones de producir una adecuada respuesta inmunitaria.
En pollos se ha inducido tolerancia a la caseína de la leche después de administrar el antígeno por vía oral. Este caso no fue posible sensibilizar al animal por vía parenteral al aplicarle la caseína (74).
En las dermatitis por contacto provocada por los metales níquel, cromo y estaño de los aretes de fantasía, se han realizado varios trabajos experimentales consistentes en inducir tolerancia por vía oral administrando dosis mínimas del metal por esta vía. Procedimiento que impidió el establecimiento de la dermatitis por contacto, cuando se trató intencionalmente de sensibilizar con estos metales, por vía cutánea (4, 12, 45).
En este momento se trabaja en la obtención de tolerancia inmunitaria por vía oral y sublingual en los procesos alérgicos a drogas; ejemplos exitosos de esta terapia se tienen los siguientes:
– En la obtención de tolerancia a la trimetoprin sulfa en pacientes con SIDA que resultaron alérgicos a este antimicrobiano (147).
– En pacientes alérgicos a penicilinas y, en general, a antibióticos con el anillo beta lactámico (141).
Como una conclusión de la revisión que hecha sobre los elementos moleculares, humorales y celulares que se ponen de manifiesto en las respuestas de inmunotolerancia loca, según que se ha descubierto hasta la fecha, se sugiere el siguiente perfil de laboratorio:
- Interleucinas : TGF-b1, IL-4,IL-10.
- Células que se activan: LTh2, y LTh3 (175).
Referencias Bibliográficas
- 1. Guerra R. Specific Antibodies in Mouse Milk after Ovalbumina Ingestion. Departamento de Biología. Universidad Federal de Maranhao, Sao Luis, Brasil. Braz-J-Med-Biol-Res, 1991; 24(1): 107-109.
- 2. Whitacre-CC, Giennapp-IE, Orosz-CG, Bitar-DM. Oral tolerance in experimental autoinmune encephalomyelitis III. Evidence for clonal anergy. Department of medical microbiology and immunology,Ohio State,University College of medicine, Columbus 43-210. J.Immunol.1991 oct. 1; 147(7): 2155-63.
- 3. Thurau-Srñ Chan-CC, Suh E, Nussemblat RB. Induction to oral tolerance to s-antigen induced experimental autoinmune uveitis by uveitogenic 20mer peptide. Laboratory of Immunology National Eye Institute, NHI, Bethesda, MD 20892.
- 4. Van Hoogstraten-IM, Burrows D, Camarasa JG, Dooms-Goossens A, Kraal G, Lahti A, et al. Reduced frecuency of nickel allergy upon oral nickel contact at an early age Department of Pathology Free University Amsterdam, The Netherlands. Clin-exp-Immunol,1991 Sep; 85 (3): 441-5.
- 5. Brod SA; Sabbagh A, Sobel RA, Hafler DA, Weiner HL. Suppression of experimental autoinmune encephalomyelitis by oral administration of Myelin antigen; IV.Suppression of chronic relapsin disease in the Lewis rat and strain 13 guinea pig. Department of Medicine, Brighams and Women’s Hospital, Boston, MA 02115. Ann-Neurol, 1991 Jun; 29(6): 615-22.
- 6. Hejjaoui J, Ferrando R, Drivert H, Michel FB, Bousquet J. Systemic reactions occurring during immunotherapy with standardized pollen extracts. J Allergy Clin Immunol. 1992;89: 925-33.
- 7. Tabar AL, García BE, Rodríguez A, Olaguibel JM, Muro MD, Quirce S. A prospective safety-monitoring study of immunotherapy with biologically standardized extracts. Allergy 1993;48: 450-3.
- 8. García Tamayo F, Fierro L, Rivera R. Oral tolerance impairment in mice with staphylococci – induced wasting syndrome. Departamento de Biología, Facultad de Química de la Universidad Nacional Autónoma de México, D.F. Arch- med – res, Spring l 992; 23(1): 33-7.
- 9. Meaga J, Fugihashi K, Yamamoto M, et al. Cytokine production and T cell receptor expression by salivary glands T cells and intraepithelial T Lymphocites for the regulation of the Ig A response. Department of oral Biology, University of Alabama, Birminham. Adv- Exp – med- Biol, 1992; 327: 119- 31.
- 10. Brandtzaeg P, Halstensen TS, Huitfeldt HS, et al. Epithelial expression of HLA, secretory component (poly Ig.) Receptor, and adhesion molecules in the human alimentary tract. Laboratory for Immunohistochemistry and Immunopathology (LIIPAT), University of Oslo, National Hospital, Rikshospitalat, Norway, Ann-N-Y-Acad Scl, 1992; 664: 157-79.
- 11. Pierre P, Denis O, Bazin H, Mbongolo Mbella E, Vaerman JP. Modulation of oral tolerance to ovalbumin by cholera toxin and its B subunit. UCL-ICP- MEXP, Brussels, Belgyum. Eur-J- Immunol. 1992 Dec; 22(12): 3179-82.
- 12. Van-Hoogstraten IM, Bodenj D, Vonj-Blomberg ME, Kraal G, Scheper RJ. Persistent immune tolerance to nickel and chromium by oral administration prior to cutaneous sensitization. Department of Pathology, Viraje University, Amsterdam, The Netherlands. J-Invest-Dermatol. 1992 Nov; 99(5): 608-16.
- 13. Revillard JP, Cozon G, Czerkinski C. Oral administration of immunomodulators and the mucosal immune system. Laboratoire d’Immunologie, INSERM U80 CNRS URA 1177 UCBL.Hopital E. Herriot, Lyon, France. Dev-Biol-Stand.1992; 77: 31-7.
- 14. Tlaskalova-Hogenova-H, Mandel L, Stepankova R, et al. Autoimmunity: from physicology to pathology. Natural antibodies, mucosal immunity and development of B cell repertoire. Division of Immunology and Gnotobiology, Czehoslovak academi of Sciences, Praha. Folia-Biol-Praha. 1992; 38 (3-4): 202-15.
- 15. Pescovitz MD. Organ acceptance and rejection. Department of surgery, Indiana University Hospital, Indianapolis 46202. Curr-Opin-Ímmunol. 1992 Oct; 4 (5): 577-81.
- 16. Hancock WW, Weiner HL. Oral Tolerance to myelin basic protein and natural recoveriy from experimental autoimmune encephalomyelitis are associated with downregulation of imflammatory cytokines and differential upregulation of transformin growth factorb1, interleukin 4, and prostaglandin E expression in the brain. Khoury SJ; Department of Medicine, Brigham and women’s Hospital Harvard Medical School, Boston, Massachusetts 02115. J-Exp-med. 1992 nov1; 176 (5): 1355-64.
- 17. Keren DF. Antigen processing in the mucosal immune system.Warde medical Laboratory, Ann Arbor, MI 48108. Semin- immunol. 1992 Aug; 4 (4): 217-26.
- 18. Zettel L, Clark DA, Ernst PB. Transforming growth factor-b1 enhances Ig.G and Ig.A sheep red blood cell responses. Molecular Virology and Immunology program, McMaster University, Hamilton, Ontario, Canadá. Int-Immulol. 1992 sept; 4-(9): 975-83.
- 19. Fujihashi K, Taguchi T, Aicher WK, et al. Immunoregulatory functions for murine intraepithelial lymphocytes: gd T Cell receptor – Positive (TCR+) T Cells abrogate oral tolerance, while ab TRC + T Cells provide B Cell help. Departament of Oral Biology, University of Alabama, Birmingham 35294. J. Exp. Med. 1992 Mar 1; 175 (3): 695-707.
- 20. Sayegh MH, Khoury SJ, Hancock WW, Weiner HL, Carpenter CB. Laboratory Induction of immunity and Oral tolerance with polymorphic class II major histocompatibility complex allopeptides in the rat. Of Immunogenetics and Transplantation, Brigham and women’s Hospital, Harvard Medical School, MA 02115. Proc- Natl. Acad. Sci. U.S.A. 1992 Aug 15; 89 (16): 7762-6.
- 21. Elson CO. Cholera toxin as a mucosal adjuvant: effects of H-2 major histocompatibility complex and lps genes. Division of Gastroenterology, University of Albama, Birmingham 35294. Infect- Immun. Jul; 60 (7): 2874-9.
- 22. Clarke CJ, Stokes C. Rallu administtered antigens in liposom: I, the response to liposome – The Internal Thadani-U and serum humoral immune response of mice to systemically And. entrapped soluble proteins. Departament of Veterinary Medicine, University of Bristol, Langford, UK. Vet- Immunol- Immunopathol. 1992 Apr; 32 (1-2): 125-38.
- 23. González Ariki S, López MC, Roux ME. IgA B lymphocytes and subpopulations of T lymphocytes in the Intestinal villi of immunodeficient wistar rats. Departament of Biological Sciences, Faculty of Pharmacy and Biochemistry, Buenos Aires University, Argentina. Reg-Immunol. 1992 Jan- Feb; 4 (1): 41-5.
- 24. Lockey RF, Benedict LM, Turkeitaub PC, Bukantz SC. Fatalities from immunotherapy and skin testing, J Allergy Clin Immunol 1987; 79: 660-77.
- 25. Umesaki Y, Setoyama H. Immune responses of mice to orally administered asialo GMI- Specific rabbit IgG in the presence or absence of cholera toxin. Yakult Central Institute for Microbiological Research, Tokyo, Japan. Immunology. 1992 Feb; 75 (2): 386-8.
- 26. Rothschild BM. Oral tolerance and Collagen arthritis (Letter; Comment) (See Comments) Br. J. Rheumatol. 1992 Mar; 31 (3): 213-4.
- 27. Muir A, Schatz D, Maclaren N. Antigen- Specific immunotherapy; Oral tolerance and subcutaneous immunization in the treatment of insulin – dependent diabetes. Departament of pathology and Laboratory Medicine, University of Florida, Gainesville. Diabetes. Metad. Rev. 1993 Dec; 9 (4): 279-87.
- 28. Ishii N, Moriguchi N, Sugita Y, Nakajima H, Tanaka S, Aoki. Analysis of responsive Cells in tolerance by the oral administration of ovalbumin. Departament of Dermatology, Yokohama City University School of Medicine, Japan. Immunol. Invest. 1993 Aug- Oct; 22 (6 -7): 451-62.
- 29. Revillard JP. (Recent advances in Physiopathology of autoimmune diseases and their therapeutic implications). Laboratoire d’ immunologie, INSERM U 80, hopital Edouard- Herriot, Lyon, France. Pediatrie. 1993; 48 (11): 759- 69.
- 30. Thompsom HS, Harper N, Bevan DJ, Staines NA. Suppression of collagen induced arthritis by oral administration of type II collagen: Changes in immune and arthritic responses. Mediated by active peripheral suppression. Infection & Immunity group, king’ s Collage London, U.K.. Autoimmunity. 1993; 16 (3): 189-99.
- 31. Melamed D, Friedman A. Modification of the immune response by oral tolerance: antigen requirements and interaction with immunogenic stimuli. Departament of Animal Sciences, Hebrew University of Jerusalem, Rehovot, Israel. Cell – Immunol. 1993 Feb; 146-20.
- 32. Aroeira LG, Carvalho CR, Mengel J, García G, Vaz NM. Hydroxyurea before oral antigen blocks the induction of oral tolerance. Departamento de Bioquímica e Inmunología, Universidade Federal de Minas Gerais, Belo Horizonte, Brasil. Braz. J. Med. Biol. Res. 1993 Oct; 26 (10): 1057-67.
- 33. Mowat AM, Maloy KJ, Donachie AM. Immune-Stimulating complexes as adjuvants for inducing local and Systemic immunity after oral immunization with protein antigens. Departament of Immunology, University of Glasgow, U.K. Immunology. 1993 Dec; 80 (4): 527- 34.
- 34. Verdolin BA, Faria AM, Carvalho CR, Lahmann WM, Vaz NM. Systemic immunization of mature mice by the oral route. Departamento de Bioquímica e Inmunología, Universidade Federal de Minas Gerias, Belo Horizonte, Brasil. Braz. J. Med. Biol. Res. 1993 Jul; 26 (7): 725-34.
- 35. Suh ED, Vistiva BP, Chan CC, Raber JM, Gery I, Nussenblatt RB. Splenectomy abrogate the induction of oral tolerance in experimental autoimmune uveoretinitis. Laboratory of Immunology, National Eye Institute, National Institutes of Health, Bethesda, MD 20892. Curr. Eye. Res. 1993 Sep; 12 (9): 833-9.
- 36. Wang ZY, Qiao J, Melms A, Link H. T Cell reactivity to acetylcholine receptor in rats orally tolerized against experimental autoimmune myasthenia gravis. Departeament of Neurology, Karolinka Institute, Huddinge Hospital, Stockholm, Sweden. Cell- Immunol. 1993 Dec; 152 (2): 394- 404.
- 37. Metzler B, Wraith DC. Inhibition of experimental autoimmune encephalomeyitis by inhalation but not oral administration of the encephalitogenic peptide: influence of MHC binding affinity. Cambrindge University Department of pathology, UK. Int- Immunol. 1993 Sep; 5 (9): 1159- 65.
- 38. Gregerson DS, Obritsch WF, Donoso LA. Oral tolerance in experimental autoimmune uveoretinitis. Distinct mechanisms of resistance are induce by low dose vs. high dose feeding protocolos. Departament of Ophthalmology, University of Minnesota, Minneapolis 55455. J. Immunol. 1993 Nov 15; 151 (10): 5751-61.
- 39. Kagnoff MF. Immunology of the intestinal tract. Departaments of Medicine, University of California, San Diego, La Jolla. Gastroenterology. 1993 Nov; 105 (5): 1275-80.
- 40. Laissue JA, Chappuis BB, Muller C, Reubi JC, Gebbers JO. The intestinal immune System and relation to disease. Institute of pathology, University of Bern, Switzerland. Dig. Dis. 1993; Jul-Oct; 11 (4-5): 298-312.
- 41. Weiner HL. Treatment of autoimmune diseases by oral tolerance to autoantigens. Enter for Neurologic Diaseases, Brigham and women’s Hospital. Harvad Medical School, Boston, MA 02115. Autoimmunity. 1993; 15 Suppl: 6-7.
- 42. Hanson DG, Roy MJ, Green GM, Miller SD. Inhibition of orally- induced immune tolerance in mice by prefeeding an endopeptidase inhibitor. Departament of pediatrics, Harvard Medical School, Boston, MA. Reg. Immunol. 1993 mar- Apr; 5 (2): 76- 84.
- 43. Miller A, Zhang ZJ, Sobel RA, Al Sabbagh A, Weiner HL. Suppression of experimental autoimmune encephalomyelitis by oral administration of myelin basic protein VI. Suppression of adoptively transferred disease and differential effects of oral vs.Intravenous tolerization. Departament of Medicine, Brigham and women’ s Hospital, Boston, MA. J. Neuroimmunol. 1993 Jul; 46 (1-2): 73-82.
- 44. Voltz R, Hohlfeld R. Current therapy of multiple sclerosis. Oral tolerance (induction to myelin antigen). Neurologische klinik, Ludwiing. Maximiliaans. Universitat, klinikum Grosshadern, Munchen. Nervenarzt. 1993 Jun; 64 (6): 411-2.
- 45. Van Hoogstraten IM, Boos C, Boden D, Von-Blomberg ME, Scheper RJ, Kraal G. Oral induction of tolerance to nickel sensitization in mice. Departament of pathology, Vrije Universiteit, Amsterdam, the Netherlands. J. Invest. Dermatol. 1993 Jul; 101 (1): 26-31.
- 46. Kim SM, Enomoto A, Hachimura S, Yamauchi K, Kaminogawa S. Serum antibody response elicted by a casein diet is directed to only limited determinants of a Sl- Casein. Departament of Agricultural Chemistry, University of Tokyo, Japan. Int. Arch. Allergy. Immunol. 1993; 101 (3): 260-5.
- 47. Maclaren NK. Diabetes intervention therapy. Departament of pathology and Laboratory Medicine, University of Florida Collage of Medicine, Gainesville 32610. J- Diabetes- Complications. 1993 Apr- Jun; 7 (2): 151-6.
- 48. Blumberg RS, Yockey CE, Gross GG, Ebeet EC, Balk SP. Human intestinal intraepithelial Lymphocytes are derived from a limited number of T Cell Clones that utilize multiple Vb T Cell receptor genes. Gastroenterology Division, Brigham and women’ s Hospital, Harvard Medical School, Boston, MA 02115. J. Immunol. 1993 Jun 1: 5144- 53.
- 49. Hoyne GF, Callow MG, Kuhlman J, Thomas WR. T. Cell Lymphokine response to orally administered proteins during priming and unresponsiveness. Division of Molecular Biology, Western Australian Research Institute for Child health, Princess Margaret Hospital, Perth.Immunology. 1993 Apr; 78 (4): 534- 40
- 50. Liu LM, Macpherson GG. Antigen acquisition by dendritic cell: intestinal dendritic cell acquire antigen administered orally and can prime naive T cell in vivo. Sir william Dunn School of Pathology, University of Oxford, Unted Kingdom. J. Exp. Med. 1993 May 1; 177 (5): 1299- 307.
- 51. Melamed D, Friendman A. Direct evidence for anergy in T Lymphocytes tolerized by oral administration of ovalbumin. Departament of Animal Science, Hebrew University of Jesusalem, Rehovot, Israel. Eur. J. Immunol. 1993 Apr; 23 (4): 935- 42.
- 52. Enomoto A, Konishi M, Hachimura S. University of Tokyo, Japan. Milk whey protein fed as a constituent of the diet induced both oral tolerance and a systemic humoral response, while heat- denatured whey protein induced only oral tolerance. Clin. Immunol. Immunopathol. 1993 Feb; 66 (2): 136-42.
- 53. Sayedg MH, Khoury SJ, Hancock WW, Weiner HL, Carpenter CB. Induction of immunity and oral tolerance to alloantigen by polymorphic class II major histocompatibility complex allopeptides in the rat. Medical School, Boston, MA 02115. Transplant. Proc. 1993 Feb; 25 (1 pt 1): 357- 8.
- 54. Faria AM, García G, Ríos MJ, Michalaros CL, Vaz NM. Decrease in susceptibility to oral tolerance induction and occurrence of oral immunization to ovalbumin in 20-38 -week- old mice. The effect of interval beetween oral exposures and rate of antigen intake in the oral immunization. Departamento de Microbiologia, Instituto de Ciencias Biologicas, UFMG, Belo Horizonte. Brasil. Immunology. 1993 Jan; 78 (1): 147-51.
- 55. Taurdorf E, Laursen L, Lanner A, Bjorkstén B, Dreborg S, Weeke B. Specific IgE, IgG, and IgA antibody response to oral immunotherapy in birch pollinosis. J Allergy Clin immunol 1989;83: 589-94.
- 56. Kaminogawa S, Hisatsune T, Hachimura S. (The molecular biology of oral tolerance). Departament of applied Biological Chemistry, University of Tokyo, Japan. Tanpakushitsu. Kakusan. Koso. 1994 Sep; 39 (12): 2090-101.
- 57. Rizzo LV, Miller Rivero NE, Chan CC, Wiggert B, Nussenblatt RB, CASPI RR. Interleukin- 2 treatment potentiates induction of oral tolerance in a murine model of autoimmunity. Laboratory of Immunology, National Eye Institute, Bethesda, Maryland 20892. J. Clin. Invest. 1994 Oct: 1668- 72.
- 58. Aramaki Y, Fujii Y, Suda H, Suzuki I, Yadomae T, Tsuchiya S. Induction of oral tolerance after feeding of ragweed pollen extract in mice. Tokyo Collage of pharmacy, Japan. Immunol. Lett. 1994 Apr; 40 (1): 21- 5.
- 59. Chen Y, Kuchroo VK, Inobe J, Hafler DA, Weiner HL. Regulatory t cell clones induced by oral tolerance: Suppression of autoimmune encephalomyelitis. Departament of medicine, Brigham and women‘s Hospital, Harvard Medical School, Boston, MA 02115. Science. 1994 Aug 26; 265 (5176): 1237- 40.
- 60. Al Sabbagh A, Miller A, Santos LM, Weiner HL. Antigen. driven tissue. specific suppression following oral tolerance: oraly administered myelin basic protein suppresses proteolipid. Protein- induced experimental autoimmune encephalomyelitis in the SJL mouse. Center for Neurologic Diseases, Brigham and women’s Hospital, Boston, MA 02115. EUR. J. Immunol. 1994 Sep: 2104-9.
- 61. Migita K, Ochi A. Induction of Clonal anergy by oral administration of Staphylococcal enterotoxin b. Departament of Immunology a Medical Genetics, University of Toronto. Eur. J. Immunol. 1994 Sep; 24 (9): 2081- 6.
- 62. Melamed D, Friedman A. In vivo tolerization of Th 1 limphocytes following a single feeding with ovalbumin; anergy in the absence of suppression. Departament of Animal Sciences, Hebrew University of Jerusalem, Rehovot, Israel. Eur. J. Immunol. 1994 Sep; 24 (9): 1974- 81.
- 63. Santos LM, Al- Sabbagh A, Londono A, Weiner HL. Oral Tolerance to myelin basic protein induces regulatory. TGF-b1- secreting T Cells in Peyer’s patches of SJL mice. Departament of Medicine, Brigham and Women’s Hospital, Boston. Massachusetts 02115. Cell. Immunol. 1994 Sep; 157 (2): 439- 47.
- 64. Wang ZY, Link H, Ljungdahl A, et al. Induction of g interferon-, interleukin- 4, and transforming growth factor-b1 in rats orally tolerized against experimental autoimmune myasthenia gravis. Departament of Neurology, Karolinska Institute, Huddinge University Hospital, Stockolm, Sweden. Cell. Immunol. 1994 Sep; 157 (2): 353- 68.
- 65. Stok W, Van Der, Heijden PJ, Bianchi AT. Conversion of orally induced suppression of the mucosal immune response to ovalbumin into stimulation by conjugating ovalbumin to cholera toxin or its b subunnit. Departament of Immunology, Central Veterinary Institute, Lelystad, the Netherlands. Vaccine. 1994 May; 12 (6): 52- 6.
- 66. Sieper J, Mitchison AN. (Therapy with oral type II collagen as a new possibility of selective immunosuppression in therapy of rheumatoid arthritis). Deutsches Rheuma. Forschungszentrum Berlin. Z- Rheumatol. 1994 Mar- Apr; 53(2): 53-8.
- 67. Friedman A, Weiner HL. Induction of anergy or active suppression following oral tolerance is determined by antigen dosage. Center for Neurologic Diseases, Brigham and women‘s Hospital, Harvard Medical school. Boston, MA 02115. Proc. Natl. Acad. Sci. U. S. A. 1994 Jul 5; 91 (14): 6688- 92.
- 68. Miller BG, Whittemore CT, Stokes CR, Telemo E. The effect of delayed wening on the development of oral tolerance to soya- bean protein in pigs. School f Veterinary Science, Bristol University, Langford. Br. J. Nutr. 1994; 71(4): 615-25.
- 69. Weiner HL, Friedman A, Miller A, Khoury SJ, Al Sabbagh A, Santos L. Oral tolerance: immunologic mechanisms and treatment of animal and human antigens. Center for Neurologic Diseases, Brigham and women’s Hospital, Boston, Massachusetts. Annu. Rev. Immunol. 1994; 12: 809- 37.
- 70. Friedman A, Al Sabbagh A, Santos LM, et al. Oral tolerance: a biologically relevant pathway to generate peripheral tolerance external and self antigens. Center for Neurological Diseases, Brigham and women’s Hospital, Boston, Mass. Chem. Immunol. 1994; 58: 259- 90.
- 71. Carvalho CR; Verdolin BA, De Souza AV. Indirect effects of Oral tolerance in mice Instituto de Ciencias Biologicas, Universidade Federal de Minas Gerais Belo Horizonte, Brasil.Scand. J. Immunol. 1994 Jun; 39 (6): 533- 8.
- 72. Hancock WW, Khoury SJ, Carpenter CB, Sayegh MH. Differential effects of oral versus intrathymic administration of polymorphic major histocompatibility complex class II Peptides on mononuclear and endothelial cell activation and cytokine expression during a delayed- type hypersensitivity response. Departament of pathology and Immunology, Monash Medical Schooll. Alfred Hospital, Prahran, Victoria, Australia. Am. J. Pathol. 1994 Jun; 144 (6): 1149- 58.
- 73. Miller A, Lider O, Abramsky O, Weiner HL. Orally administered myelin basic protein in neonates primes for immune responses And enhances experimental autoimmune encephalomyelitis in adult animal. Departament of Neurology, Carmel Medical Center, Haifa, Israel. Eur. J. Immunol. 1994 May; 24 (5): 1026-32.
- 74. Miller CC, Cook ME. Evidence against the induction of immunological tolerance by feeding antigens to chickens. Departament of poultry Science, University of Wisconsin- Madison 53706. Poult- Sci. 1994 Jan; 73 (1): 106-12.
- 75. Husby S, Mestcky J, Moldoveanu Z, Holland S, Elson CO. Oral tolerance in humans. T Cell but not B cell tolerance after antigen feeding. Departament of medicine. University of Alabama at Birmingham 35294. J. Immunol. 1994 May 1; 152 (9): 4663- 70.
- 76. Hoyne GF, Callow MG, Kuo MC, Thomas WR. Differences in epitopes recognized by T cells during Oral Tolerance and priming. Division of Molecular Biology, Western Australian Research Institute For Chid Health, Princess Margareth Hospital, Perth. Immunol. Cell. Biol. 1994 Feb; 72(1): 29-33.
- 77. Feliziani V, Marfisi RM, Parmiani S. Rush immunotherapy with sublingual administration of grass allergen extract. Allergol Immunopathol (Madr) 1993;21: 173-8.
- 78. Van Hoogstraten IM, Von Blomberg BM, Boden D, Kraal G, Scheper RJ. Non-sensitizing epicutaneous skin test prevent subsequent induction of immunotolerance. Deapartament of pathology, Vrije Universiteit, Amsterdam, the Nestherdans. J. Invest. Dermacol. 1994 Jan; 102 (1): 80.
- 79. Hafler Da, Weiner HL. Antigen-specific immunosuppression: Oral tolerance for the treatment of autoimmune diseases. Laboratory of Molecular Immunology, Center for Neurologic Diseases, Brigham and Women’s Hospital, Boston, Mass. Chem. Immunol. 1995; 60: 128-49.
- 80. Yoshino S. (Suppression of autoimmune diseases by induction of oral tolerance to self antigen). Nippon. Saikingaku. Zasshi. 1994 Sep; 49(5-6): 737-44.
- 81. Van Wilsem EJ, Van Hoogstraten IM, Breve J, Scheper RJ, Kraal G. Dendritic cells of the oral mucosa and the induction of oral tolerance. A local affair. Departament of cell Biology & Immunology, Vrije Universiteit, Amsterdam, The Netherlands. Immunology. 1994 Sep; 83(1): 128-32.
- 82. Takahashi I, Nakagawa I, Kiyono H, McGree JR, Clements JD, Hamada S. Mucosal T cells induce systemic anergy for oral tolerance. Departament of oral Microbiology, Osaka University Faculty of Dentistry, Japan. Biochem. Biophys. Res. Commun. 1995 Jan 5; 206 (1): 414-20.
- 83. Matsui M, Kuroda Y. (Gut mucosal immunity and oral tolerance). Departament of Internal Medicine, Saga Medical School. Nippon. Rinsho. 1994 Nov; 52 (11): 2873-9.
- 84. Weiner HL. Oral tolerance (comment). Center for Neurologic Diseases, Brighm and Women’s Hospital, Boston,MA 02115. Proc-Natl. Acad. Sci. U. S. A. 1994 Nov 8; 91 (23): 10762-5.
- 85. Yang R, Liu Q, Grosfeld JL, Pescovitz MD. Intestinal venous drainage through the liver is a prerequisite for oral tolerance induction. Departament of Surgery, Indiana University School of Medicine, Indianapolis. J. Pedriatr. Surg. Aug 1994 ; 29(8): 1145-8.
- 86. Carr DJ, Woolley TW, Clarke BL. The relationship between the hypothalamic-pituitary adrenal axis and oral-induced immunosuppression. Departament of Microbiology, Immunology, and Parasitology, Louisiana State University Medical Center, New Orleans 70112. Int. J. Neurosci. 1994 May; 76(1-2): 151-64.
- 87. Fishman Lobell J, Friedman A, Weiner HL. Different kinetic patterns of cytokine gene expression in vivo in orally tolerant mice. Center for Neurological Diseases, Brigham and Women’s Hospital, Harvard Medical School, Boston,MA 02115. Eur. J. Immunol. 1994 Nov; 24(11): 2720-4.
- 88. MacDonald TT. Oral tolerance. Eating your way towards immunosuppression. Departament of Pediatric Gastroenterology, Medical Collage of St Bartholomews Hospital, London, UK. Curr-Biol. 1994 Feb 1; 4(2): 178-81.
- 89. Matsumoto Miike T, Ono T. Mite- related allergy and oral tolerance. Departament of Child Development Kumamoto University School of Medicine. Japan. Allergy. 1996 Apr; 51 (4): 276- 7.
- 90. Vistica BP, Chanaud NP, Felix N, et al. CD8 T-cell are not essential for the induction of “Low – dose” oral tolerance Laboratory of Immunology, National Eye Institute, NIH, Berthesda, Maryland 20892. Clin. Immunol. Immunopathol, 1996 Feb; 78 (2): 196-202.
- 91. Nussenblatt RB, Whitcup SM, De Smet MD, et al. Intraocular imflamatory disease (uveitis) and the use of oral tolerance; a status report. Laboratory of Immunology, National Eye Institute, National Institutes of Health. Bethesda, Maryland 20892, USA. Ann. N. Y. Acad. Sci. 1996 Feb 13; 778: 325- 37.
- 92. Fujihashi K, McGree JR, Yamamoto M, Hiroi T, Kiyono H. c d T cells in Role of the regulation of mucosal IgA response – and oral tolerance. Departament of Oral Biology, University of Alabama at Birmingham, 35294, USA. Ann. N. Y. Acad. Sci. 1996 Feb 13; 778: 55- 63.
- 93. Yuan YS, Major JA, Battisto JR. Regulation of Chemokine gene expression by contact hypersensibility and by oral tolerance. Departament of Biology, Cleveland State University, Ohio 44195, USA. Ann. N. Y. Acad. Sci. 1996 Feb 13; 778: 434- 7.
- 94. Bour H, Nicolas JF, Kaiserlian D. In serum U 404, Immunity et Vaccination, Institute Pasteur, Lyon, France. Lack of oral tolerance but oral priming for contact sensitivity to dinitrofluorobenzene in major histocompatibility complex class II- deficient mice and LTh4 cell. depleted mice. Desvingnes. C; Eur. J. Immunol. 1996 Aug; 26 (8): 1756-61.
- 95. Van Houten N, Blaske SF. Direct measuremet of anergy of antigen. specific T cells following oral tolerance induction. Departament of Internal Medicine, Galveston 77555-0366, USA. nvanhout@beanch.utmb.edu. Immunol. 1996 Aug 15; 157 (4): 1337- 41.
- 96. Sun JB, Rask C, Olsson, T, Holmgren J, Czerkinsky C. Treatment autoimmune encephalomyelitis by feeding myelin basic of experimental protein conjugated To cholera toxin, B subunit. Departament of Medical Microbiology and Immunology- University of Goteborg, Sweden. Proc. Natl. Acad. Sci. U. S. A. 1996 Jul 9; 93 (14): 7196-201.
- 97. Fukaura H, Kent SC, Pietrusewiez MJ, Kroury SJ, Weiner HL, Hafler DA. Induction of circulating myelin basic protein and proteolipid protein – specific transforming growth factorb1-secreting TH3 cells by oral administration of myelin in multiple sclerosis patients. Center for Neurologic Diseases, Deapartament of Neurology, Bringm and women’s Hospital, Boston, Massachusetts, USA. J. Clin. Ivest. 1996 Jul 1; 98 (1): 70-7.
- 98. Neurath MF, Fuss I, Kelsall BL, Presky DH, Waegell W, Strober W. Experimental granulomatous colitis in mice is abrogated by induction of TGFb1 mediated oral tolerance. Mucosal Immunity Section, National Institutes of Health, NIH/NIAID/LCI, Bethesda, Maryland 20892. 1890, USA. J. Exp. Med. 1996 Jun 1; 183 (6): 2605- 16.
- 99. Kagnoff MF. Oral tolerance mechanisms and possible role in imflammatory joint diseases. Laboratory of Mucosal Immunology, University of California, San Diego, La Jolia 92093-0623, USA. Baillieres. Clin. Rheumatol. 1996 Feb; 10 (1): 41-54.
- 100.Repoport B, McLachlan SM. Food for thought is induction of oral tolerance feasible and practical in human thyroid autoimmunity? (editorial comment). Comment on: Endocrinology 1996 Jun; 137 (6): 2199-207. Endrocrinology. 1996 Jun; 137 (6): 2197- 8.
- 101.Palacios As, medina FS, Rua-Figueroa Al, Millares CB, Santos SR, Marrero JAG, Comparative clinical-immunological study of oral and subcutaneous immunotherapy in children with extrinsic bronchial astma. Allergol Immunopathol (Madr) 1989;17: 323-9.
- 102.Nelken D (Sublingual immunotherapy). Asthma and Allergy Clinic Center, Tel Aviv, Harefauh, 1996 Sep; 131 (5-6): 164-5,215.
- 103.Lundin BS, Dahlgren UI, Hanson LA, Telemo E. Oral tolerization leads to active suppression and bystander tolerance in adult rats while anergy dominate in young rats. Departament of Clinical Immunology, Goteborg University, Gothenburg, Sweden. Scand- J. Immunol. 1996 Jan; 43 (1): 56-63.
- 104.Ke Y, Kapp JA. Oral antigen inhibits priming of CD8+CTL, CD4+T cell, and antibody response while activating CD8+ suppressor T cells. Departament of pathology, Emory University School of Medicine, Atlanta, GA 30322, USA. J. Immunol. 1996 Feb 1; 156 (3): 916-21.
- 105.Chen Y, Inobe J, Kuchroo VK, Baron JL, Janeway CA Jr, Weiner HL. Oral tolerance in myelin basic protein T- cell receptor transgenics mice: Suppression of autoimmune encephalomyelitis and dose, dependent induction of regulatory cells. Center for Neurologic Diseases, Brigham and Women’s Hospital, Harvard Medical School, Boston, MA 02115, USA. Proc. Natl. Acad. U.S.A. 1996 Jan 9; 93 (1): 388-91.
- 106.Arakawa Y, Suzuki K, Oyama R, Moriyama M. *Encephalomyelitis -Allergic immunology; *Myelin – basic – proteins -genetics; *Receptors, Antigen, T. Cell. Physiology. Animal; Suppor,. U.S. Gov‘t,. P.H.S. JOURNAL-ARTICLE.
- 107.Litwin A, Flanagan M, Entis G, et al. Oral immunotherapy with short ragweed extract in a novel encapsulated preparation; a double-blind study, J. Allergy. Clin. Immunol. 1997;100: 30-8.
- 108.Ke Y, Kapp JA. Oral antigen inhibits priming of CD8+CTL, CD4+T cells, and antibody responses while activating CD8+ suppressor T cells. Departament of Pathology, Emory University School of Medicine, Atlanta, GA 30322, USA. J. Immunol. 1996 Feb 1; 156(3): 916-21.
- 109.Brandizaeg P. History of oral tolerance and mucosal immunity. Laboratory for Immunohistochemistry and Immunopathology, Institute of Pathology, University of Oslo, Norway. Ann. N. Y. Acad. Sci. 1996 Feb 13; 778: 1-27.
- 110.Piazza I, Bizzaro N. Humoral response to subcutaneus, oral,and nasal immunotherapy for allergic rhinitis due to Dermatophagoides pteronyssinus, Ann Allergy 1993;71: 461-9.
- 111.Weiner HL, Mayer LF. Oral tolerance; mechanisms and appications, Introduction. Center for Neurologic Diseases, Brigham and women’s Hospital, Boston, Massachussets 02115, USA. Ann. N. Y. Acad. Sci. 1996 Feb 13; 778: xii-xvii.
- 112.Mowat AM, Steel M, Worthey EA, Kewin PJ, Garside P. Inactivation of Th1 and Th2 cells by feeding ovalbumin. Departament of Immunology, University of Glasgow, Scotland.. Ann. N. Y. Acad. Sci. 1996 Feb 13; 778: 122-32.
- 113.Chen YH, Weiner HL. Dose-dependent activación and deletion of antigen – specific T cells following oral tolerance. Center for Nurological diseases, Departament of medicine, Harvard Medical School, Boston, Massachusetts 02115, USA. Ann. N. Y. Acad. Sci. 1996 Feb 13; 778: 111-21.
- 114.Friedman A. Induction of anergy in Th1 lymphocytes by oral tolerance. Importance of antigen dosage and frequency of feeding. Departament of Animal Sciences, Hebrew University of Jerusalem, Rehovot, Israel. Ann. N. Y. Acad. Sci. 1996 Feb 13; 778: 103-10.
- 115.Nelson PA, Akselband Y, Dearborn SM, et al. Effect of oral b interferon on subsequent immune responsiveness. Autoimmune Inc. Lexington, Massachussetts 02173, USA. Ann. N. Y. Acad. Sci. 1996 Feb 13; 778: 145- 55.
- 116.Karpus WJ, Lukacs NW. The role of chemokines in oral tolerance. Abrogation of nonresponsiveness by treatment with antimonocyte chemotactic protein-l. Departament of Microbiology and Immunology, Nortwesterm University Medical School, Chicago, Ilinois 60611, USA. Ann. N. Y. Acad. Sci. 1996 Feb 13; 778: 143-44.
- 117.Metzler B, Wraith DC. Mucosal tolerance in a murine model of experimental autoimmune encephalomyelitis. Division of Immunology, Departament of Pathology, Cambridge University, United Kingdom. Ann. N. Y. Acad. Sci. 1996 Feb 13; 778: 228-42.
- 118.Whitacre CC, Gienapp IE, Meyer A, Cox KL, Javed N. Oral tolerance in experimental autoimmune encephalomyelitis. Departament of Medical Microbiology and Immunology, Ohio State University College of Medicine,Columbus, 43210, USA. Ann. N. Y. Acad. Sci. 1996 Feb 13; 778: 217-27.
- 119.Caspi RR, Stiff LR, Morawetz R,et al. Morse-HC-3ard, Rizzo-LV. Cytokine – dependent modulation of oral tolerance in a murine model of autoimmune uveitis. Laboratory of Immunology, National Eye Institute, Bethesda, Maryland 20892, USA. Ann. N. Y. Acad. Sci. 1996 Feb 13; 778: 315-24.
- 120.Staines NA, Harper N, Ward FJ, Thompson HS, Bansal S. Arthritis; animal models of oral tolerance. Infection and Immunity Research Group, Division of Life Science, King’s Collage London, United Kingdom. Ann. N. Y. Acad. Sci. 1996 Feb 13; 778: 297-305.
- 121.Drachman DB; Okmura S; Adams RN; McIntosh KR. Oral tolerance in myasthenia gravis. Departament of Neurology, Johns Hopkins University, School of Medicine, Baltimore, Maryland 21287-7519, USA.. Ann. N. Y. Acad. Sci. 1996 Feb 13; 778: 258-72.
- 122.Flanagan M, Jin S, Michael JG. Braking of oral tolerance by encapsulated antigen. Departament of Molecular Genetics, Collage of Medicine, University of Cincinnati, Ohio 45212-0524, USA. Ann. N. Y. Acad. Sci. 1996 Feb 13; 778: 373-5.
- 123.Maron R, Blogg NS, Polanski M, Hancock W, Weiner HL. Oral tolerance to insulin b-Chain; Cell Iines and cytokine patterns. Center for Neurologic Diseases, Harvard Medical School, Boston, Massachusetts 02115,USA. Ann. N. Y. Acad. Sci, 1996 Feb 13; 778: 346-57.
- 124.Sayedh MH, Khoury SJ, Hancock WW, Weiner HL, Carpenter CB. Mechanisms of oral tolerance by MHC Peptides. Laboratory of Immunogenetics and Transplantation, Harvard Medical School, Boston, Massachusetts 02115, USA. Ann. N. Y. Acad. Sci, 1996 Feb 13; 778: 338-45.
- 125.Meyer A, Gienapp I, Cox K, Goverman J, Hood L, Whitacre C. Oral tolerance in myelin basic protein TCR transgenics mice. Departament of Medical Microbiology and Immunology, Ohio State University Collage of Medicine, Columbus 43210, USA. Ann. N. Y. Acad. Sci. 1996 Feb; 778: 412-3.
- 126.Javed NH, Giennapp I, Cox K, Whitacre CC. Oral tolerance in experimental autoimmune encephalomyelitis: specificity of peptide induced oral tolerance. Departament of Medical Microbiology and Immunology, Ohio State University, Collage of Medicine, Columbus 43210, USA. Ann. N. Y. Acad. Sci. 1996 Feb; 778: 393-4.
- 127.Gienapp I, Cox K, Javed N, Whitacre CL, Isola S. Oral tolerance in autoimmune encephalomyelitis in vivo reversal of anergy.
- 128.D’Ambrosio FP, Ricciardi Puccinelli P, Musarra A. Rush sublingual immunotherapy in parietaria allergic patients. Policlinico Universitario di Messina, Scuola di specializzazione in Allergologia ed Inmunología Clínica, Milano, Italia. Allergol. Immunopathol. Madr. 1996 Jul-Aug; 24(4): 146-51.
- 129.Montgomery PC, Skandera CA, O‘Sullivan NL. Fenotypic profile of lymphocyte population isolated from rat major salivary glands. Departament of Immunology and Microbiology, Waye State University School of Medicine, Detroit, Michigan 48201, USA. Oral. Microbiol. Immunol. 1996 Aug; 11 (4): 248-53.
- 130.Quirino T, Iemoli E, Siciliani E, Parmiani S, Milazzo F. Sublingual versus injective inmmunotherapy in grass pollen allergic patients: a double blind (double dummy) study (see comments). Comments in: Clin. Exp. Allergy 1996 Nov; 26 (11): 1228-31. Servizio di Allergologia Ospedale L. Sacco, Milan, Italy. Clin. Exp. Allergy. 1996 Nov; 26 (11): 1253-61.
- 131.Malling- HJ. sublingual immunotherapy (editorial. Comment on; Clin Exp Allergy 1996 Nov; 26 (11): 1253-61. Clin. Exp. Allergy. 1996 Nov; 26(11): 1228-31.
- 132.Garside P, Mowat AM. Mechanisms of oral tolerance. Departament of Immunology, University of Glasgow. Crit. Rev. Immunol. 1997; 17 (2): 119-37.
- 133.Gimsa U, Sieper J, Braun J, Mitchison NA. Type II Collage serology; a guide to clinical responsiveness to oral tolerance?. Dautsches Rheuma. Forschungs. Zentrum. Berlin, Germany. Rheumatol-Int. 1997; 16 (6): 237-40.
- 134.Carvalho CR, Verdolin BA, Vaz NM. Indirect effects of oral tolerance cannot be ascribed to bystander suppression. Instituto de Ciencias Biologicas, Universidade Federal de Minas Gerais, Belo Horizonte, Brazil. Scand. J. Immunol. 1997 mar; 45 (3): 276-87.
135.Yoshida T, Hachimura S, Kaminogawa S. The oral administration of low-dose antigen induces activation followed by tolerization, while high-dose antigen induces tolerance without activation. Departament of Applied Biological Chemistry, University of Tokyo, Japan. Clin. Immunol. Immunopathol. 1997 Mar; 82 (3): 201-15. - 136.Singh VK, Nagaraju K. Experimental autoimmune uveitis; molecular mimicry and oral tolerance. Departament of Immunology, Sanjay Gandhi Post-Graduate Institute of Medical Sciences, Lucknow, Indiana. Immunol. Res. 1996; 15(4): 323-46
- 137.Melamed D, Fishman Lovell J, Uni Z, Weiner HL, Friedman A. Peripheral tolerance of Th2 lymphocytes induced by continuos feeding of ovalbumin. Departament of Animal Sciences, Hebrew University of Jerusalem, Rehovot Israel. Int. Immunol. 1996 May; 8(5): 717-24.
- 138.Kaminogawa S. Food allergy, oral tolerance and immunomodulation-their molecular and cellular mechanisms. Departament of Applied Biological Chemistry, University of Tokyo, Japan. Biosci-Biotechnol-Biochem. 1996 Nov; 60(11): 1749-56.
- 139.Van Alteren AG, Van der Cammen MJ, Biewenga J, Savelkoul HF, Kraal G. Ig E and Mast cell response on intestinal allergen exposure: a murin model to study the onset of food allergy. Department of Cell Biology and Immunology, Vrije Universiteit Amsterdam, The Netherlands. J. Allergy Clin. Immunology. 1997 Jan; 99 ( 1 Pt 1): 94-9.
- 140.Inada S, Yoshino S, Haque MA, Ogata Y, Kohashi O. Clonal anergy is a potent mechanism of oral tolerance in the suppression of accute antigen-induced arthritis in rats by oral administration of the inducing antigen. Departament of Microbiology, Saga Medical School, Japant. Cell. Immunol. 1997 Jan 10; 175(1): 67-75.
- 141.Gamboa P, Jáuregui I, Urrutia I, González G, Antepara I. Contact sensitization to cloxacillin with oral tolerance to other b lactam antibiotics. Section de Alergia-Inmunología, Hospital de Basurto, Avda, Bilbao, Spani. Contact. Dermatitis. 1996 Jan; 34(1): 75-6.
- 142.Harper HM, Cochrane L, Williams NA. The role of small intestinal antigen-presenting cell in the induction of T-cell reactivity to soluble protein antigens; association between aberrrant presentation in the lamina propria and oral tolerance. University of Bristol, Departament of pathology and Microbiology, School of Medical Sciences, University Walk, UK. Immunology. 1996 Nov; 89(3): 449-56.
- 143.Haque MA, Yoshino S, Inada S, Nomaguchi H, Tokunaga O, Kohaschi O. Suppression of adjuvant arthritis in rats by induction of oral tolerance to mycobacterial 65-kDa heat shock protein. Departament of Microbiology, Saga Medical School, Japan. Eur. J. Immunol. 1996 Nov; 26(11): 2650-6.
- 144.Marth T, Strober W, Kelsall BL. High dose oral tolerance in ovalbumin TCR-trangenic mice; systemic neutralization of IL-12 augments TGF-b secretion and T cell apoptosis. Laboratory of Clinical Investigation, National Institute of Allergy and Infectious Diseases, Bethesda, MD 20892, USA. J. Immunol. 1996 Sep 15; 157(6): 2348-57.
- 145.Kelly KA, Whitacre CC. Oral tolerance in EAE; reversal of tolerance by T helper cell cytokines. Departament of Microbiology and Immunology, University of Arkansas for Medical Sciences, Little Rock 72205, USA. J. Neuroimmunol. 1996 May; 66(1-2): 77-84.
- 146.Matsui S, Yokomuro-Mabuchi A, Tanaka S. Characteristic immunological tolerance in intrahepatic lymphocytes induced by bacterial superantigen SEB. Departament of Microbiology and Immunology, Nippon Medical School, Tokyo, Japan. Nippon-Ika-Daigaku. Zasshi. 1996 Feb; 63 (1): 22-30.
- 147.Kalanadhabhatta V, Muppidi D, Sahni H, Robles A, Kramer M. Successful oral desensitization to trimethoprim-sulfamethoxazole in acquired immune deficiency syndrome. Division of Allergy/Clinical Immunology, Departament of Medicine, Medicine state University of New York, Health Sciences Center at Brooklyn,USA.. Ann. Allergy. Astma. Immunol. 1996 Nov; 77(5): 394-400.
- 148.Holt PG, Britten D, Sedgewick JD. Suppression of IgE responses by antigen inhalation: Studies on the role of genetic and environmetal factors. Immunology. 1987: 60: 97-102.
- 149.Van Wilsem EJ, Van Hoogstraten IM, Breve J, Sheper RJ, Kraal G. Dendritic cell of the oral mucosa and the induction of oral tolerance. A local affair Immunology. 1994; 83: 128-32.
- 150.Hasseus B, Dahlgren U, Bergenholz G, Jontell M. Antigen presenting capacity of Langerhams cell from rat oral epithelium. Oral Pathol Med. 1995; 24: 56-60.
- 151.Manetti R, Parronchi P, Giudizi MG, et al. Natural Killer cell stimulatory factor interleukin 12 (IL- 12) induces T helper type 1 (Th1) –specific immune responses and inhibits the development of IL-4- producing TH cell. Exp. Med. 1993; 177: 1199-1204.
- 152.Macatinia SE, Hosken NA, Litton M, et al. Dendritic cells produce Il- 12 and direct the development of TH1 cell from naive CD4+T cell. J Immunol. 1995; 154: 5071-9.
- 153.Bagnasco M, Mariani G, Passalacqua G, et al. Absorption and distribution kinetics of the major Parietaria Judaica, allergen administered by noninjectable routes to healthy human beings. J Allergy Clin. Immunol. 1997; 100: 122-9.
- 154.Greenberg MA Kaufman CR, González GE. Late and immediate systemic allergic reactions to inhalant allergen immunotherapy. J Allergy Clin. Immunol. 1986; 77: 865-70.
- 155.Giovane Al, Bardare M, Passalacqua G, et al. A three- year double- blind placebo- controlled study with specific oral immunotherapy to Dermatophagoides: evidence of safety and efficacy in Pediatric patients. Clin. Exp. Allergy. 1994; 24: 53-9.
- 156.Johansson SGO, Deuschl H, Zetterstrom O. Use of glutar- aldehyde- modified timothy grass pollen extract in nasal hiposensitisation treatment of hay fever. Int Arch Allergy Appl Immunol. 1979; 60: 447-60.
- 157.Andri L, Senna G, Andri G, et al. Local nasal immunotherapy for birch allergic rhinitis with extract in powder form. Clin Exp Allergy. 1995; 25: 1092-9.
- 158.Ariano R, Panzani RC, Chiapella M, Augeri G, Falagiani P. Local immunotherapy of easonal allergic rhinitis in children due to Parietaria Pollen. A preliminary report. Pediatric Asthma Allergy Immunol. 1993; 7: 227-37.
- 159.Welsh PW, Butterfield JH, Yunginger W, Agarwal MK, Gjeich GJ. Allergen controlled study of intranasal immunotherapy for ragweed hay fever. J Allergy Clin Immunol. 1983; 71: 454-60.
- 160.Crimi E, Voltolini S, Troise C, et al. Local immunotherapy with Dermatophagoide extract in asthma. J Allergy Clin Immunol. 1991; 87: 721-8.
- 161.Tari MG, Mancino M, Monti G. Immunotherapy by inhalation of allergen in powder in house dust allergic asthma; a double – blind study. J Investing Allergol Clin Immunol.1992; 2: 59-67.
- 162.Taurdorf E, Laursen LC, Djurup R, et al. Oral administration of grass pollen to hay fever patients. An efficacy study in oral hiposensitization. Allergy. 1985; 40: 321-35.
- 163.Mosbech H, Dreborg S, Madsen F, et al. Night dose grass pollen tablets use for hyposensitization in hay fever patients. A one- year double blind placebo controlled study. Allergy. 1987; 42: 451-5.
- 164.Taudorf E, Laurse LC, Lanner A, et al. Oral immunotherapy in birch pollen hay fever. Allergy Clin Immunol. 1987; 80: 153-61.
- 165.Scadding K, Brostoff J. Low dose sublingual therapy in patients with allergic rhinitis due to dust mite. Clin. Allergy. 1986; 83: 589-94.
- 166.Nelson HS, Oppenheimer J, Vatsia GA, Bucheimer A. A double blind placebo controlled evaluation of sublingual immunotherapy with standardized eat extract. J Allergy Clin. Immunol. 1993; 92: 229-36.
- 167.Casanova M, Guerra F, Moreno C, Miguel R, Maranon F, Daza JC. Double blind placebo controlled – clinical trial of preseasonal treatment with allergic extracts of Olea europea pollen administered sublingually. J Investing Allergol Clin. Immunol. 1994; 4: 305-14.
- 168.Tari MG, Mancino M, Monti G. Efficacy of sublingual immunotherapy in patients with rhinitis and asthma due to house dust mite. A double blind study. Allergol Immunopathol (Madr). 1990; 18, 277-84.
- 169.Sabbah A, Hassoun S, Le Scllin J, André C, Sicard H. A double blind placebo controlled trial by the sublingual route of immunotherapy with a standardized grass pollen extract. Allergy. 1994; 49: 309-13.
- 170.Troise C, Voltolini S, Canessa A, Pecora S, Negrini AC. Sunblingual immunotherapy in parietaria pollen induced rhinitis; a double blind study. J Investig Allergol Clin. Immunol. 1995; 5: 25-30.
- 171.Hirsch T, Sahn M, Leupoid W. Double- blind placebo- controlled study of Sunblingual immunotherapy with house dust mite extract (D, pt.) in Children. Pediatric Allergy immunol. 1997; 8: 21-7.
- 172.Passalacqua G, Albano M, Fregonese L, et al. Randomised controlled trial of local allergoid immunotherapy on allergie inflammation in mite. Induced rhinoconjuntivitis. Lancet. 1998; 351: 629-32.
- 173.Watkin AD. The role of alternative therapies in the treatment of allergic diseases. Clin. Exp. Allergy. 1994; 24: 813-25.
- 174.Malling HJ, Abreu- Nogueira J, Alvarez, Cuesta E, et al. Local immunotherapy. Allergy 1998; 53: 933-944, º Munksgard 1998.
- 175.Howard L.Weiner E. P. Rees. Mucosal tolerance. Immunology Letters. vol 69, numero1, junio15 de 1999.
- 176.A.M.C. Faria. Weiner. Oral tolerance mechanisms and therapeutics applications, Adv. Immunol. 1999; 73: 153-264.
- 177.Eilidh Williamson and Joanne L, Viney. Department of Molecular munology Immunex Corporation email: Dendrictic Cell are Key Regulators of Intestinal Imune Response. Mucosal Immunology Update. 1999; Vol. 7, Nª- 2, pág. 7.