En los países desarrollados del mundo, donde el acceso al tratamiento del CM está menos obstaculizado por barreras del sistema de salud, la mortalidad por CM ha disminuido progresivamente entre los años noventas del siglo pasado y la actualidad (9,131). En el CM metastásico con RH positivos, específicamente, la supervivencia prácticamente se ha duplicado, pasando de 32 a 57 meses (132).
Aunque esta mejoría puede ser atribuible a muchos factores, no hay duda de que el desarrollo de nuevos fármacos ha tenido un papel fundamental. Sin embargo, todavía existen importantes preguntas que contestar, particularmente en aquellas pacientes que desarrollan resistencia al tratamiento hormonal.
Las vías futuras de investigación incluyen el estudio y tratamiento de tumores con mutaciones en el RE, el hallazgo de biomarcadores, y el uso de biopsias líquidas, con la intención de desarrollar tratamientos personalizados que permitan mejorar la supervivencia de estas pacientes.
(Lea También: Perspectiva Actual de la Cirugía Oncológica)
Conclusiones
La introducción y posterior evolución de muchos agentes endocrinos desde la década de 1970 ha transformado el tratamiento de las mujeres con CM (Figura 3).
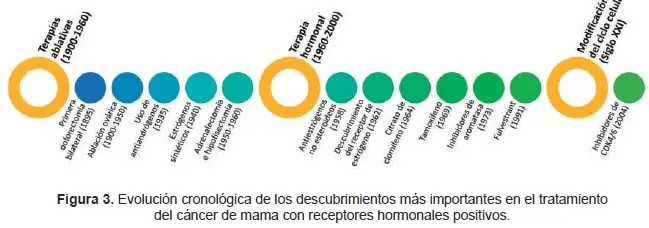
Estos avances han logrado un cambio en la práctica clínica actual, pasando de cirugías ablativas y quimioterapias agresivas, a tratamientos dirigidos y mejor tolerados, lo que ha mejorado la supervivencia y la calidad de vida de las pacientes con CM.
Conflictos de interés
Yanin Chávarri Guerra ha recibido financiamiento para investigación de Roche y apoyo de viáticos de Pfizer. Los demás autores se declaran sin conflictos de interés.
Agradecimientos
Agradecemos a la diseñadora Luí Balmonte por el diseño y elaboración de las figuras 1 y 2.
Referencias
-
1. (IARC) IA for R on C, World Health Organization (WHO). Breast Cancer. Source: Globocan 2018. World Heal Organ. 2018;876:2018-2019.
-
2. Bloom D. Breakaway: The global burden of cancer -challenges and opportunities. A report from the Economist Intelligence Unit Limited 2009. London: The Economist; 2009.
-
3. Bravo LE, García LS, Carrascal E, Rubiano J. Burden of breast cancer in Cali, Colombia: 1962-2012. Salud Publica Mex. 2014;56(5):448-456.
-
4. Soto-Perez-de-Celis E, Chavarri-Guerra Y. National and regional breast cancer incidence and mortality trends in Mexico 2001-2011: Analysis of a population-based database. Cancer Epidemiol 2016;41:24-33
-
5. Duarte C, Salazar A, Strasser-Weippl K, de Vries E, Wiesner C Krush L et al. Abstract P5-13-12: Breast cancer in Colombia: A growing challenge for the health care system. Cancer Res.:1-6.
-
6. SEER*Explorer: An interactive website for SEER cancer statistics [Internet]. Surveillance Research Program NCI [consultado 14 septiembre 2020].Disponible en: https://seer.cancer.gov/explorer/.
-
7. Villarreal-Garza C, Aguila C, Magallanes-Hoyos MC et al. Breast Cancer in Young Women in Latin America: An Unmet, Growing Burden. Oncologist. 2013;18(S2):26-34.
-
8. Waks AG, Winer EP. Breast Cancer Treatment: A Review. JAMA. 2019;321(3):288-300.
-
9. Shumway DA, Sabolch A, Jagsi R. Breast Cancer. Med Radiol. 2020:1-43. 10.
-
10. Obeidat F, Ahram M, Al Khader A et al. Clinical and histopathological features of breast cancer in Jordan: Experience from a tertiary care hospital. J Pak Med Assoc. 2017;67(8):1206-1212.
-
11. Goldhirsch A, Winer EP, Coates AS, et al. Personalizing the treatment of women with early breast cancer: Highlights of the st gallen international expert consensus on the primary therapy of early breast Cancer 2013. Ann Oncol. 2013;24(9):2206-2223.
-
12. Sainsbury R. The development of endocrine therapy for women with breast cancer. Cancer Treat Rev. 2013;39(5):507-517.
-
13. Dao TL, Huggins C. Metastatic cancer of the breast treated by adrenalectomy evaluation and the five-year results Chicago Materials and Methods The first series of patients treated ectomy , or by this operation combined with oophor- ectomy , consisted of 52 consecutive patie. JAMA. 1957;165(14):1793-1797.
-
14. Singh G. Oophorectomy in Breast Cancer-Controversies and Current Status. Indian J Surg. 2012;74(3):210-212.
-
15. Russell PW. Hypophysectomy in metastatic breast carcinoma. J Kans Med Soc. 1955;56(9):481 485.
-
16. Jensen EV, Block GE, Smith S, Kyser K DE. Estrogen receptors and breast cancer response to adrenalectomy. Natl Cancer Inst Monogr. 1971;34:55-.
-
17. Huggins C, Bergenstal DM. Inhibition of Human Mammary and Prostatic Cancers by Adrenalectomy. Cancer Res. 1952;12(2):134-141.
-
18. Friedman N, Jaffe HL, Rabwin MH, Rosenblum DH, Simkin B. Adrenalectomy for control of cancer of the breast. Calif Med. 1956;85(4):213-219.
-
19. Olivercrona RL and H. Hipophysectomy in the treatment of malignant tumors. Cancer. 1957;16(10):788-794. 20. Silverberg GD, Britt RH. Transsphenoidal hypophysectomy in the treatment of metastatic breast and prostate carcinoma. West J Med. 1979;130(3):191-195. 21. Lewison EF. The treatment of Advanced breast cancer. Am J Nurs. 62(10):107-110.
-
22. Khan S. Estrogen receptor and breast cancer: a historical perspective. Cancer Drug Discov Dev. 2019:1-14.
-
23. Allen E, Doisy EA. An Ovarian Hormone: Preliminary Report on Its Localization, Extraction and Partial Purification, and Action in Test Animals. JAMA J Am Med Assoc. 1983;250(19):2681-2683.
-
24. Doisy EA. An Autobiography. Annu Rev Biochem. 1976.
-
25. Herting T A. Allen and Doisy’s “An Ovarian Hormone.” JAMA. 1983;250(19).
-
26. Allen E, Doisy EA. An ovarian hormone. Preliminary reports on its localization, extraction and partial purification and action in test animals. J Am Med Assoc. 1923;81(10):819-821.
-
27. Doisy EA. The crystals of thefollicular ovarian hormone. Proc Soc Exp Biol Med. 1930;27(5):417-419.
-
28. Butenandt A. Über “Progynon” ein krystallisiertes weibliches Sexualhormon. Naturwissenschaften. 1929;17(879). 29.
-
29. Santen RJ, Simpson E. History of estrogen: Its purification, structure, synthesis, biologic actions, and clinical implications. Endocrinology. 2019;160(3):605-625.
-
30. Lerner LJ, Jordan VC. Development of Antiestrogens and Their Use in Breast Cancer: Eighth Cain Memorial Award Lecture. Cancer Res. 1990;50(14):4177-4189.
-
31. Russo J, Russo IH. The role of estrogen in the initiation of breast cancer. J Steroid Biochem Mol Biol. 2006;102(1-5 SPEC. ISS.):89-96.
-
32. Haddon, A.. Influence of synthetic oestrogens upon advanced malignant disease. Brit MedJ. 1944:2:393-398.
-
33. Schwander H, Marvin HN. Treatment of carcinoma of the human breast with testosterone propionate: A report of five Cases. J Clin Endocrinol Metab. 1947;7(6):423-432.34.
-
34. Sala JM, Del Regato JA. Treatment of carcinoma of the endometrium. Radiology. 1962;79:12-17.
-
35. McGraw AB. Testosterone propionate in treatment of recurrent cancer of the breast. Arch Surg. 1948;57(3):385- 390. 36.
-
36. Kennedy BJ. Massive Estrogen Administration in premenopausal women with metastatic breast cancer. Cancer. 1962;15(5):641-648.
-
37. Allen E,Doisy E. An ovarian hormone preliminary report on its localization, extraction and partial purification, and action in test animals. JAMA. 1923;81(10):819.
-
38. Macgregor JI, Jordan VC. Basic guide to the mechanisms of antiestrogen action. Pharmacol Rev. 1998;50(2):151-196.
-
39. Craig Jordan V, McDaniel R, Agboke F, Maximov PY. The evolution of nonsteroidal antiestrogens to become selective estrogen receptor modulators. Steroids. 2014;90:3-12.
-
40. Herbst AL, Griffiths CT, Kistner RW. Clomiphene citrate (NSC-35770) in disseminated mammary carcinoma. Cancer Chemother Rep. 1964;43:39-41.
-
41. Jensen E V. On the Mechanism of Estrogen Action genic activity. Perspect Biol Med .2018;6(1):47-60.
-
42. Jensen E V, Suzuki T, Kawashima T, Stumpf WE, Jungblut PW, DeSombre ER. A two-step mechanism for the interaction of estradiol with rat uterus. Proc Natl Acad Sci. 1968;59(2):632 LP-638.
-
43. Moore DD. A conversation with Elwood Jensen. Annu Rev Physiol. 2012;74:1-11.
-
44. Jensen E V., Jordan VC. The estrogen receptor: A model for molecular medicine. Clin Cancer Res. 2003;9(6):1980-1989.
-
45. Gorski J, Noteboom W. Stereospecific Binding of Estrogens in the Rat Uterus. Arc Biochem Biophy. 1965;111(3):559-568.
-
46. Toft D, Shyamala G, Gorski J. A receptor molecule for estrogens: studies using a cell-free system. Proc Natl Acad Sci U S A. 1967;57(6):1740-1743.
-
47. Greene GL, Fitch FW, Jensen E V. Monoclonal antibodies to estrophilin: Probes for the study of estrogen receptors. Proc Natl Acad Sci U S A. 1980;77(1):157-161. 48.
-
King WJ, Greene GL. Monoclonal antibodies localize oestrogen receptor in the nuclei of target cells. Nature. 1984;307(5953):745-747.49.
-
49. Krust A, Green S, Argos P, et al. The chicken oestrogen receptor sequence: homology with v-erbA and the human oestrogen and glucocorticoid receptors. EMBO J. 1986;5(5):891-897.
-
50. Kumar V, Green S, Staub A, Chambon P. Localisation of the oestradiol-binding and putative DNA-binding domains of the human oestrogen receptor. EMBO J. 1986;5(9):2231-2236.
-
51. Williams C, Lin CY. Oestrogen receptors in breast cancer: Basic mechanisms and clinical implications. Ecancermedicalscience. 2013;7(1):1-13. 52.
-
52. Kuiper GGJM, Enmark E, Pelto-Huikko M, Nilsson S, Gustafsson JÅ. Cloning of a novel estrogen receptor expressed in rat prostate and ovary. Proc Natl Acad Sci U S A. 1996;93(12):5925-5930.
-
53. Todd IDH. Prediction of Response in Cancer Therapy. Br J Cancer. 1972;26(5):423-423.
-
54. Jensen E V. Hormone dependency of breast cancer. Cancer. 1981;47(10):2319-2326.
-
55. Maass H, Engel B, Hohmeister H, Lehmann F, Trams G. Estrogen receptors in human breast cancer tissue. Am J Obstet Gynecol. 1972;113(3):377-382.
-
56. Persijn JP, Korsten CB. Oestrogen Receptor in Human Breast Cancer Tissue and Response to Endocrine Therapy. Br Med J. 1973;2(5869):750.
-
57. Criss WE, Bland KI, O’Leary JP. Predictability of response to endocrine ablation in breast cancer. South Med J. 1975;68(6):714-716.
-
58. Meredith JT, McBride RC, Cerezo L. Estrogen receptors in breast cancer. J Fla Med Assoc. 1988;75(1):22-28.
-
59. Jensen E V. Estrogen Receptors in Hormone-Dependent Breast Cancers. Cancer Res. 1975;35:3362-3364.
-
60. Kaye AH, Laws ER. Historical Perspective. Brain Tumors. 2012:1-5.
-
61. Kwakowsky A, Milne M, Waldvogel H, Faull R. Effect of Estradiol on Neurotrophin Receptors in Basal Forebrain Cholinergic Neurons: Relevance for Alzheimer’s Disease. Int J Mol Sci. 2016;17:2122.
-
62. Saji S, Jensen E V, Nilsson S, Rylander T, Warner M, Gustafsson JA. Estrogen receptors alpha and beta in the rodent mammary gland. Proc Natl Acad Sci U S A. 2000;97(1):337-342.
-
63. Platet N, Cathiard AM, Gleizes M, Garcia M. Estrogens and their receptors in breast cancer progression: A dual role in cancer proliferation and invasion. Crit Rev Oncol Hematol. 2004;51(1):55-67.
-
64. Saji S, Hirose M, Toi M. Clinical significance of estrogen receptor β in breast cancer. Cancer Chemother Pharmacol. 2005;56(SUPPL. 7):21-26.
-
65. Weigel NL. Steroid hormone receptors and their regulation by phosphorylation. Biochem J. 1996;319(3):657-667.
-
66. Rong C, Fasolt É, Corvin R, Hess J. Estrogen Receptor Signaling in Radiotherapy : From Molecular Mechanisms to Clinical Studies. 2018.
-
67. Katzenellenbogen BS. Estrogen receptors: Bioactivities and interactions with cell signaling pathways. Biol Reprod. 1996;54(2):287-293.
-
68. O’Malley BW. A life-long search for the molecular pathways of steroid hormone action. Mol Endocrinol. 2005;19(6):1402-1411. doi:10.1210/me.2004-0480.
-
69. Girgert R, Emons G, Gründker C. Estrogen signaling in ERα-negative breast cancer: ERβ and GPER. Front Endocrinol (Lausanne). 2019;10(JAN):1-12.
-
70. Klinge CM. Estrogens regulate life and death in mitochondria.J Bioenerg Biomembr 2017;49(4):307-324
-
71. Liao TL, Lee YC, Tzeng CR, et al. Mitochondrial translocation of estrogen receptor β affords resistance to oxidative insult-induced apoptosis and contributes to the pathogenesis of endometriosis. Free Radic Biol Med. 2019;134(October 2018):359-373.
-
72. Vona R, Ascione B, Malorni W, Straface E. Mitochondria and Sex-Specific Cardiac Function. In: Advances in Experimental Medicine and Biology. 2018:241-256.
-
73. Perillo B, Sasso A, Abbondanza C, Palumbo G. 17β-Estradiol Inhibits Apoptosis in MCF-7 Cells, Inducing bcl-2 Expression via Two Estrogen-Responsive Elements Present in the Coding Sequence. Mol Cell Biol. 2000;20(8):2890-2901.
-
74. Engelbreth-Holm J. Acceleration of the Development of Mammary Carcinomas in Mice by Methylcholanthrene. Cancer Res. 1941;1(2):109-112.
-
75. Bielschowsky F. The carcinogenic action of 2-acetylaminofluorene and related compounds. Br Med Bull. 1946;4(5-6):382-384. 76.
-
76. Bielschowsky F. Distant tumours produced by 2-aminoand 2-acetylamino-fluorene. Br J Exp Pathol. 1944;25(1).
-
77. Geyer RP, Bryant JE, Bleisch VR, Peirce EM, Stare FJ. Effect of Dose and Hormones on Tumor Production in Rats Given Emulsified 9,10-Dimethyl-l,2-benzanthracene Intravenously. Cancer Res. 1953;13:503-506.
-
78. Shay H. Development of adenocarcinoma of the breast in the wistar rat following the gastric instillation of methylcholanthrene. J Natl Cancer Inst. 1949;10(2):255-266.
-
79. Cantarow A, Stasney J, Paschkis KE. The lnfluence of Sex Hormones on Mammary Tumors Induced by 2-Acetaminofluorene. Cancer Res. 1948;8(9):412-417.
-
80. Huggins C, Morii S, Grand LC. Mammary cancer induced by a single dose of polynuclear hydrocarbons: routes of administration. Ann Surg. 1961;154(6):315-318.
-
81. Walsh PC. How Charles Huggins made his nobel prize winning discovery-in his own words: An historic audio recording. Prostate. 2012;72(16):1718-1718.
-
82. Harper K, Walpole AL. A New Derivative of triphenilethylene: effect on implatation and mode of action in rats. J Reprod Fert. 1966;13:101-119.
-
83. Klopper A, Hall M. New Synthetic Agent for the Induction of Ovulation: Preliminary Trials in Women. Br Med J. 1971;1(5741):152-154.
-
84. Jordan VC. A current view of tamoxifen for the treatment and prevention of breast cancer. Br J Pharmacol. 1993;119:507-517.
-
85. Jordan VC. Antiestrogenic and antitumor properties of tamoxifen in laboratory animals. Cancer Treat Rep. 1976;60(10):1409-1419.
-
86. Satyaswaroop PG, Zaino RJ, Mortel R. Estrogen-like Effects of Tamoxifen on Human Endometrial Carcinoma Transplanted into Nude Mice. Cancer Res. 1984;44(9):4006-4010.
-
87. Jordan VC, Koerner S. Tamoxifen (ICI 46,474) and the human carcinoma 8S oestrogen receptor. Eur J Cancer. 1975;11(3):205-206.
-
88. Cole MP, Jones CTA, Todd IDH. A new anti-oestrogenic agent in late breast cancer an early clinical appraisal of ICI46474. Br J Cancer. 1971;25(2):270-275.
-
89. Jordan VC. Tamoxifen: A most unlikely pioneering medicine. Nat Rev Drug Discov. 2003;2(3):205-213.
-
90. Breast E, Trialists C, Group C. Polychemotherapy for early breast cancer: an overview of the randomised trials. Early Breast Cancer Trialists’ Collaborative Group. Lancet . 1998;352(9132):930-942.
-
91. Vogel VG, Costantino JP, Wickerham DL, Cronin WM. Re: Tamoxifen for prevention of breast cancer: Report of the National Surgical Adjuvant Breast and Bowel Project P-1 Study [1]. J Natl Cancer Inst. 2002;94(19):1504.
-
92. Cuzick J. First results from the International Breast Cancer Intervention Study (IBIS-I): A randomised prevention trial. Lancet. 2002;360(9336):817-824.
-
93. Jordan VC. Tamoxifen as the first targeted long-term adjuvant therapy for breast cancer. Endocr Relat Cancer. 2014;21(3):1-20.
-
94. Santen RJ, Brodie H, Simpson ER, Siiteri PK, Brodie A. History of aromatase: Saga of an important biological mediator and therapeutic target. Endocr Rev. 2009;30(4):343-375.
-
95. Ghosh D, Griswold J, Erman M, Pangborn W. Structural basis for androgen specificity and oestrogen synthesis in human aromatase. Nature. 2009;457(7226):219-223.
-
96. Hemsell DL, Grodin JM, Brenner PF, Siiteri PK, Macdonald PC. Plasma precursors of estrogen. II. Correlation of the extent of conversion of plasma androstenedione to estrone with age. J Clin Endocrinol Metab. 1974;38(3):476-479.
-
97. Grodin JM, Siiteri PK, MacDonald PC. Source of estrogen production in postmenopausal women. Obstet Gynecol Surv. 1973;28(9):654-656.
-
98. Schwarzel WC, Kruggel WG, Brodie HJ. Studies on the Mechanism of Estrogen Biosynthesis. VIII. The Development of Inhibitors of the Enzyme System in Human Placenta. Endocrinology. 1973;92(3):866-880.
-
99. Coombes RC, Goss P, Dowsett M, Gazet JC, Brodie A 1984 4-Hydroxyandrostenedione in treatment of postmenopausal patients with advanced breast cancer. Lancet 1984;2(8414)::1237–1239.
-
100. Steele RE, Mellor LB, Sawyer WK, Wasvary JM, Browne LJ 1987 In vitro and in vivo studies demonstrating potent and selective estrogen inhibition with the nonsteroidal aromatase inhibitor CGS 16949A. Steroids. 1987;50(1-3):147–161.
-
101. Trunet PF, Mueller P, Girard F, Aupetit B, Bhatnagar AS, Zognbi F, Ezzet F, Menard J 1992 The effects of fadrozole hydrochloride on aldosterone secretion in healthy male subjects. J Clin Endocrinol Metab 74:571–576. 1992:1992.
-
102. Buzdar AU, Robertson JFR, Eiermann W, Nabholtz JM. An overview of the pharmacology and pharmacokinetics of the newer generation aromatase inhibitors anastrozole, letrozole, and exemestane. Cancer. 2002;95(9):2006- 2016. 103.
-
103. Janni W, Hepp P. Adjuvant aromatase inhibitor therapy: Outcomes and safety. Cancer Treat Rev. 2010;36(3):249- 261.
-
104. Behan LA, Amir E, Casper RF. Aromatase inhibitors for prevention of breast cancer in postmenopausal women: A narrative review. Menopause. 2015;22(3):342-350.
-
105. Lai AC, Crews CM, Haven N. Induced protein degradation. Nat Rev Drug Discov. 2017;16(2):101-114.
-
106. Carlson RW. The history and mechanism of action of fulvestrant. Clin Breast Cancer. 2005;6(SUPPL. 1):S5.
-
107. Robertson JFR, Jiang Z, Di Leo A, et al. A meta-analysis of clinical benefit rates for fulvestrant 500 mg vs. alternative endocrine therapies for hormone receptor- positive advanced breast cancer. Breast Cancer. 2019;26(6):703-711.
-
108. Sutherland RL, Musgrove EA. Cyclins and breast cancer. J Mammary Gland Biol Neoplasia. 2004;9(1):95- 104.
-
109. Buckley MF, Sweeney KJ, Hamilton JA, Sini RL, Manning DL, Nicholson RI, deFazio A, Watts CK, Musgrove EA SR. Expression and amplification of cyclin genes in human breast cancer. Oncogene. 1993;8(8):2.
-
110. Asghar U, Witkiewicz AK, Turner NC, Knudsen ES. The history and future of targeting cyclin-dependent kinases in cancer therapy. Nat Rev Drug Discov. 2015;14(2):130- 146.
-
111. Bartkova J, Lukas J, Müller H, Lützhøt D, Strauss M, Bartek J. Cyclin D1 protein expression and function in human breast cancer. Int J Cancer. 1994;57(3):353-361.
-
112. Sedlacek HH, Czech J, Naik R, et al. Flavopiridol (L86 8275; NSC 649890), a new kinase inhibitor for tumor therapy. Int J Oncol. 1996;9(6):1143-1168.
-
113. Mita MM, Joy AA, Mita A, et al. Randomized phase II trial of the cyclin-dependent kinase inhibitor Dinaciclib (MK- 7965) vs. capecitabine in patients with advanced breast cancer. Clin Breast Cancer. 2014;14(3):169-176.
-
114. Fry DW, Harvey PJ, Keller PR, et al. Specific inhibition of cyclin-dependent kinase 4/6 by PD 0332991 and associated antitumor activity in human tumor xenografts. Mol Cancer Ther. 2004;3(11):1427-1437.
-
115. Toogood PL, Harvey PJ, Repine JT, et al. Discovery of a potent and selective inhibitor of cyclin-dependent kinase 4/6. J Med Chem. 2005;48(7):2388-2406.
-
116. Rader J, Russell MR, Hart LS, et al. Dual CDK4/CDK6 inhibition induces cell-cycle arrest and senescence in neuroblastoma. Clin Cancer Res. 2013;19(22):6173- 6182.
-
117. Zhang YX, Sicinska E, Czaplinski JT, et al. Antiproliferative effects of CDK4/6 inhibition in CDK4-amplified human liposarcoma in vitro and in vivo. Mol Cancer Ther. 2014;13(9):2184-2193
-
118. Gelbert LM, Cai S, Lin X, et al. Preclinical characterization of the CDK4/6 inhibitor LY2835219: In-vivo cell cycle-dependent/independent anti-tumor activities alone/ in combination with gemcitabine. Invest New Drugs. 2014;32(5):825-837.
-
119. Tate SC, Cai S, Ajamie RT, et al. Semi-mechanistic pharmacokinetic/pharmacodynamic modeling of the antitumor activity of LY2835219, a new cyclin-dependent kinase 4/6 inhibitor, in mice bearing human tumor xenografts. Clin Cancer Res. 2014;20(14):3763-3774.
-
120. Christine Desmedt CS. The most prominent predictor of clinical outcome in breast cancer. Cell Cycle. 2006;(April):853-858.
-
121. Khera R, Kondamudi N, Zhong L, Vaduganathan M, Parker J, Das SR. Temporal Trends in Heart Failure Incidence Among Medicare Beneficiaries Across Risk Factor Strata , 2011 to 2016. JAMA Netw. 2020;3(10):1-13.
-
122. Santen RJ, Manni A, Harvey H, Redmond C. Endocrine Treatment of Breast Cancer in Women.Endocr Rev. 1990;11(2):221-65.
-
123. Shim W, Conaway M, Masamura S, Yue W, Wang J, Kmar R et al. Estradiol Hypersensitivity and Mitogen- Activated Protein Kinase Expression in Long-Term Estrogen Deprived Human Breast Cancer Cells in Vivo .Endocrinology. 2000;2000;141(1):396-405.
-
124. Barone I, Cui Y, Herynk M, Corona-Rodriguez A, Giordano C, Selever J et al. Expression of the K303R Estrogen Receptor α Breast Cancer Mutation Induces Resistance to an Aromatase Inhibitor via Addiction to the PI3K/Akt Kinase Pathway.Cancer Res. 2009;69(11):4724-4732.
-
125. Merenbakh-Lamin K, Ben-Baruch N, Yeheskel A, Dvir A, Soussan-gutman L, Jeselsohn R et al. D538G Mutation in Estrogen Receptor- a : A Novel Mechanism for Acquired Endocrine Resistance in Breast Cancer.Cancer Res. 2013;73(23):6856-6865.
-
126. Jeselsohn R, Yelensky R, Buchwalter G, Frampton G, Meric-Bernstam F, Gonzalez-Angulo A et al. Emergence of Constitutively Active Estrogen Receptor- a Mutations in Pretreated Advanced Estrogen Receptor – Positive Breast Cancer.Clin Cancer Res. 2014;20(7):1757-1767.
-
127. Toy W, Shen Y, Won H, Green B, Sakr R, Will M et al. ESR1 ligand-binding domain mutations in hormoneresistant breast cancer.Nat Genet 2013;45(12):1439- 1445.
-
128. Robinson DR, Wu YM, Vats P,Su F, Lonigro R, Cao X et al. Activating ESR1 mutations in hormone-resistant metastatic breast cancer. Nat Genet. 2013;45(12):1446- 1451.
-
129. Harrod A, Fulton J, Nguyen VTM, Periyasamy M, Ramos- García L, Lai C et al. Genomic modelling of the ESR1 Y537S mutation for evaluating function and new therapeutic approaches for metastatic breast cancer. Oncogene. 2017;36(16):2286-2296.
-
130. Toy W, Weir H, Razavi P, Lawson M, Goeppert A, Mazzola A et al. Activating ESR1 Mutations Differentially Affect the Effi cacy of ER Antagonists. Cancer Discov. 2017;7(3):277-287.
-
131. Webb PM, Cummings MC, Bain CJ, Furnival CM. Changes in survival after breast cancer: Improvements in diagnosis or treatment?. Breast. 2004;13(1):7-14.
-
132. Caswell-Jin JL, Plevritis SK, Tian L, Cadham CX, Stout NK, Sledge G et al. Change in Survival in Metastatic Breast Cancer with Treatment Advances: Meta- Analysis and Systematic Review. JNCI Cancer Spectr. 2018;2(4):1-10.
Recibido: Octubre 27, 2020
Aceptado: Noviembre 03, 2020
Correspondencia:
Yanin Chávarri Guerra
yaninchg@gmail.com